LBF18302HP02
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA8064 |
| LipidMaps | LMFA01040048 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18302HP02 |
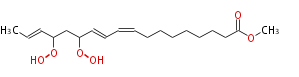
| Methyl-13,15-dihydroperoxy- (9Z,11E,16E) -octadecatrienoic acid | |
|---|---|
| |
| Structural Information | |
| Methyl-13,15-dihydroperoxy- (cis-9,trans-11,trans-16) -octadecatrienoic acid | |
| |
| Formula | C19H32O6 |
| Exact Mass | 356.219888756 |
| Average Mass | 356.45378 |
| SMILES | CC=CC(OO)CC(OO)C=CC=CCCCCCCCC(=O)OC |
| Physicochemical Information | |
| It is produced from 13- or 15-hydroperoxy isomer during oxidation of linoleate by singlet-oxygen Frankel_EN Neff_WE et al. Neff_WE et al.. | |
| It reacts with DNA in the presence of Fe ions and ascorbic acid Fujimoto_K et al.. | |
| Spectral Information | |
| Mass Spectra | GC-EI-MS(after reduction and TMS-derivatization) Neff_WE et al.: m/e=468[M]; 453[M-OCH3]; 437[M-OCH3]; 378[M-HOTMS]; 311[SMTO=CHCH=CHCH=CH(CH2)7COOCH3]; 143[SMTO=CHCH=CHCH3] |
| UV Spectra | Conjugated diene: λ max=234-235nm Neff_WE et al. |
| IR Spectra | OOH group: 3700-3140cm-1[bonded], 3520-3510cm-1[free]; conjugated cis, trans diene: 985-979cm-1, 953-935cm-1; isolated trans unsaturation: 968-960cm-1 Neff_WE et al. |
| NMR Spectra | 1H-NMR Neff_WE et al.: C2: 2.31-2.32ppm; C8, 14: 1.8-2.2ppm; C9, 10, 12, 16, 17: 5.25-6.20ppm; C11: 6.63ppm; C13, 15: 4.48-4.50ppm; C18: 1.8ppm; OOH: 7.94-8.08ppm Neff_WE et al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|