LBF15306CV02: Difference between revisions
No edit summary |
No edit summary |
||
| Line 5: | Line 5: | ||
|LipidMaps=LMFA03120026 | |LipidMaps=LMFA03120026 | ||
|SysName= (E) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde | |SysName= (E) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde | ||
|Common Name=&&clavirin II&& | |Common Name=&&clavirin II&& (E) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde&& | ||
|Reflactive=[<FONT FACE="Symbol">a</FONT>]<SUB><FONT SIZE=-1>D</FONT></SUB> -33.7°(C 0.43, CHCl<SUB><FONT SIZE=-1>3</FONT></SUB>)[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | |Reflactive=[<FONT FACE="Symbol">a</FONT>]<SUB><FONT SIZE=-1>D</FONT></SUB> -33.7°(C 0.43, CHCl<SUB><FONT SIZE=-1>3</FONT></SUB>)[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | ||
|NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(500MHz,CDCl<SUB><FONT SIZE=-1>3</FONT></SUB>)<FONT FACE="Symbol">d</FONT>ppm0.88(3H,t,J=7.1Hz),1.20-1.40(6H,m),1.95(2H,brq,J=7.2Hz),2.09(3H,s),2.88(1H,dd,J=7.1,14.5Hz),2.91(1H,dd,J=7.5,14.5Hz),5.23(1H,ddd,J=1.6,7.2,10.9Hz),5.62(1H,ddd,J=7.1,7.5,10.9Hz),6.53(1H,d,J=8.0Hz),6.54(1H,d,J=6.2Hz),7.65(1H,d,J=6.2Hz),10.33(1H,d,J=8.0Hz).[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] <SUP><FONT SIZE=-1>1</FONT></SUP><SUP><FONT SIZE=-1>3</FONT></SUP>C-NMR(125MHz,CDCl<SUB><FONT SIZE=-1>3</FONT></SUB>)<FONT FACE="Symbol">d</FONT>ppm14.0,21.3,22.5,27.5,28.9,31.4,37.3,84.4,120.1,125.6,134.8,136.5,149.4,159.3,169.2,190.6,192.5.[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | |NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(500MHz,CDCl<SUB><FONT SIZE=-1>3</FONT></SUB>)<FONT FACE="Symbol">d</FONT>ppm0.88(3H,t,J=7.1Hz),1.20-1.40(6H,m),1.95(2H,brq,J=7.2Hz),2.09(3H,s),2.88(1H,dd,J=7.1,14.5Hz),2.91(1H,dd,J=7.5,14.5Hz),5.23(1H,ddd,J=1.6,7.2,10.9Hz),5.62(1H,ddd,J=7.1,7.5,10.9Hz),6.53(1H,d,J=8.0Hz),6.54(1H,d,J=6.2Hz),7.65(1H,d,J=6.2Hz),10.33(1H,d,J=8.0Hz).[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] <SUP><FONT SIZE=-1>1</FONT></SUP><SUP><FONT SIZE=-1>3</FONT></SUP>C-NMR(125MHz,CDCl<SUB><FONT SIZE=-1>3</FONT></SUB>)<FONT FACE="Symbol">d</FONT>ppm14.0,21.3,22.5,27.5,28.9,31.4,37.3,84.4,120.1,125.6,134.8,136.5,149.4,159.3,169.2,190.6,192.5.[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | ||
}} | }} | ||
Revision as of 00:01, 20 December 2008
| IDs and Links | |
|---|---|
| LipidBank | XPR8045 |
| LipidMaps | LMFA03120026 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF15306CV02 |
| clavirin II | |
|---|---|
| |
| Structural Information | |
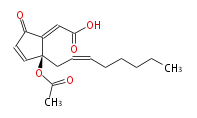
| (E) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde | |
| |
| Formula | C17H22O4 |
| Exact Mass | 290.151809192 |
| Average Mass | 290.35418 |
| SMILES | CCCCCC=CC[C@](OC(C)=O)(C=1)C(=CC=O)C(=O)C1 |
| Physicochemical Information | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | 1H-NMR(500MHz,CDCl3)dppm0.88(3H,t,J=7.1Hz),1.20-1.40(6H,m),1.95(2H,brq,J=7.2Hz),2.09(3H,s),2.88(1H,dd,J=7.1,14.5Hz),2.91(1H,dd,J=7.5,14.5Hz),5.23(1H,ddd,J=1.6,7.2,10.9Hz),5.62(1H,ddd,J=7.1,7.5,10.9Hz),6.53(1H,d,J=8.0Hz),6.54(1H,d,J=6.2Hz),7.65(1H,d,J=6.2Hz),10.33(1H,d,J=8.0Hz). IwashimaMet al. 13C-NMR(125MHz,CDCl3)dppm14.0,21.3,22.5,27.5,28.9,31.4,37.3,84.4,120.1,125.6,134.8,136.5,149.4,159.3,169.2,190.6,192.5. IwashimaMet al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|