LBF17115SC01: Difference between revisions
No edit summary |
No edit summary |
||
| Line 10: | Line 10: | ||
|Melting Point=57.5°C | |Melting Point=57.5°C | ||
|Solubility=soluble in ether / crystals from acetone[[Reference:Lauer_WM:Gensler_WJ:Miller_E:,J. Am. Chem. Soc.,1941,63,1153|{{RelationTable/GetFirstAuthor|Reference:Lauer_WM:Gensler_WJ:Miller_E:,J. Am. Chem. Soc.,1941,63,1153}}]][[Reference:Myers_GS:,J. Am. Chem. Soc.,1951,73,2100|{{RelationTable/GetFirstAuthor|Reference:Myers_GS:,J. Am. Chem. Soc.,1951,73,2100}}]][[Reference:Myers_GS:,J. Am. Chem. Soc.,1952,74,1390|{{RelationTable/GetFirstAuthor|Reference:Myers_GS:,J. Am. Chem. Soc.,1952,74,1390}}]] | |Solubility=soluble in ether / crystals from acetone[[Reference:Lauer_WM:Gensler_WJ:Miller_E:,J. Am. Chem. Soc.,1941,63,1153|{{RelationTable/GetFirstAuthor|Reference:Lauer_WM:Gensler_WJ:Miller_E:,J. Am. Chem. Soc.,1941,63,1153}}]][[Reference:Myers_GS:,J. Am. Chem. Soc.,1951,73,2100|{{RelationTable/GetFirstAuthor|Reference:Myers_GS:,J. Am. Chem. Soc.,1951,73,2100}}]][[Reference:Myers_GS:,J. Am. Chem. Soc.,1952,74,1390|{{RelationTable/GetFirstAuthor|Reference:Myers_GS:,J. Am. Chem. Soc.,1952,74,1390}}]] | ||
|Source= | |||
|Chemical Synthesis=Synthetic, by the malonic acid method from pentadecanoldehyde and malonic acid in presence of pyridine. | |||
|Metabolism= | |||
}} | }} | ||
{{Lipid/Footer}} | {{Lipid/Footer}} | ||
Revision as of 22:00, 24 November 2009
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA0098 |
| LipidMaps | LMFA01030059 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF17115SC01 |
| 2-Heptadecylenic acid | |
|---|---|
| |
| Structural Information | |
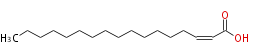
| 2-Heptadecenoic acid | |
| |
| Formula | C17H32O2 |
| Exact Mass | 268.240230268 |
| Average Mass | 268.43478 |
| SMILES | CCCCCCCCCCCCCCC=CC(O)=O |
| Physicochemical Information | |
| 57.5°C | |
| soluble in ether / crystals from acetone Lauer_WM et al. Myers_GS Myers_GS | |
| Synthetic, by the malonic acid method from pentadecanoldehyde and malonic acid in presence of pyridine. | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|