LBF18108HP04: Difference between revisions
m (LBF182nnHP01 moved to LBF18108HP04) |
No edit summary |
||
| Line 5: | Line 5: | ||
|LipidMaps=LMFA01040053 | |LipidMaps=LMFA01040053 | ||
|SysName=Methyl-11- (3,5-Epidioxy-2-Ethylcyclopentyl) -9-Hydroperoxy-10-Undecenoate | |SysName=Methyl-11- (3,5-Epidioxy-2-Ethylcyclopentyl) -9-Hydroperoxy-10-Undecenoate | ||
|Common Name=&&Methyl-11- (3,5-Epidioxy-2-Ethylcyclopentyl) -9-Hydroperoxy-10-Undecenoate&& | |||
|Mass Spectra=EI-MS(after reduction and TMS)[[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223}}]]: m/e=543[M-CH3]; 468[M-HOTMS]; 401[M-(CH2)7COOCH3]; 378[M-2xHOTMS]; 352[468-SMTO=CHCH2]; 323[352-CH2CH3]; 285[CH=CHCH(OTMS)(CH2)7COOCH3]; 259[SMTO=CH(CH2)7COOCH3]; 217[SMTO-CH=CH-CH=OTMS] | |Mass Spectra=EI-MS(after reduction and TMS)[[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223}}]]: m/e=543[M-CH3]; 468[M-HOTMS]; 401[M-(CH2)7COOCH3]; 378[M-2xHOTMS]; 352[468-SMTO=CHCH2]; 323[352-CH2CH3]; 285[CH=CHCH(OTMS)(CH2)7COOCH3]; 259[SMTO=CH(CH2)7COOCH3]; 217[SMTO-CH=CH-CH=OTMS] | ||
|IR Spectra=OOH group: 3620-3010cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[bonded], 3520cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[free]; isolated trans unsaturation: 960cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780}}]] | |IR Spectra=OOH group: 3620-3010cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[bonded], 3520cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[free]; isolated trans unsaturation: 960cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780}}]] | ||
|NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR[[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1984,106,3577|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1984,106,3577}}]][[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223}}]]: C9: 4.26-4.35; C10: 5.47-5.52; C11: 5.44-6.03; C12: 2.66-2.92; C13: 4.42-4.53; C15: 4.53-4.66; C16: 1.90-2.16; OOH: 7.89-7.94; 13C-NMR[[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1984,106,3577|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1984,106,3577}}]][[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223}}]]: C9: 86.3-86.6; C10: 129.8-132.4; C11: 132.4-135.0; C12: 45.9-50.8; C13: 79.8-80.5C14: 39.3-44.1; C15: 81.7-83.0; | |NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR[[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1984,106,3577|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1984,106,3577}}]][[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223}}]]: C9: 4.26-4.35; C10: 5.47-5.52; C11: 5.44-6.03; C12: 2.66-2.92; C13: 4.42-4.53; C15: 4.53-4.66; C16: 1.90-2.16; OOH: 7.89-7.94; 13C-NMR[[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1984,106,3577|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1984,106,3577}}]][[Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223|{{RelationTable/GetFirstAuthor|Reference:Oconnor_DE:Mihelich_ED:Coleman_MC:,J. Am. Chem. Soc.,1981,103,223}}]]: C9: 86.3-86.6; C10: 129.8-132.4; C11: 132.4-135.0; C12: 45.9-50.8; C13: 79.8-80.5C14: 39.3-44.1; C15: 81.7-83.0; | ||
}} | }} | ||
Revision as of 08:20, 19 December 2008
| IDs and Links | |
|---|---|
| LipidBank | DFA8069 |
| LipidMaps | LMFA01040053 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18108HP04 |
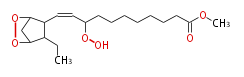
| Methyl-11- (3,5-Epidioxy-2-Ethylcyclopentyl) -9-Hydroperoxy-10-Undecenoate | |
|---|---|
| |
| Structural Information | |
| Methyl-11- (3,5-Epidioxy-2-Ethylcyclopentyl) -9-Hydroperoxy-10-Undecenoate | |
| |
| Formula | C19H32O6 |
| Exact Mass | 356.219888756 |
| Average Mass | 356.45378 |
| SMILES | C(C2CC)(O1)CC(C2C=CC(OO)CCCCCCCC(=O)OC)O1 |
| Physicochemical Information | |
| Spectral Information | |
| Mass Spectra | EI-MS(after reduction and TMS) Oconnor_DE et al.: m/e=543[M-CH3]; 468[M-HOTMS]; 401[M-(CH2)7COOCH3]; 378[M-2xHOTMS]; 352[468-SMTO=CHCH2]; 323[352-CH2CH3]; 285[CH=CHCH(OTMS)(CH2)7COOCH3]; 259[SMTO=CH(CH2)7COOCH3]; 217[SMTO-CH=CH-CH=OTMS] |
| UV Spectra | |
| IR Spectra | OOH group: 3620-3010cm-1[bonded], 3520cm-1[free]; isolated trans unsaturation: 960cm-1 Neff_WE et al. |
| NMR Spectra | 1H-NMR Oconnor_DE et al. Oconnor_DE et al.: C9: 4.26-4.35; C10: 5.47-5.52; C11: 5.44-6.03; C12: 2.66-2.92; C13: 4.42-4.53; C15: 4.53-4.66; C16: 1.90-2.16; OOH: 7.89-7.94; 13C-NMR Oconnor_DE et al. Oconnor_DE et al.: C9: 86.3-86.6; C10: 129.8-132.4; C11: 132.4-135.0; C12: 45.9-50.8; C13: 79.8-80.5C14: 39.3-44.1; C15: 81.7-83.0; |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|