LBF18303HP03: Difference between revisions
No edit summary |
No edit summary |
||
| Line 5: | Line 5: | ||
|LipidMaps=LMFA01040015 | |LipidMaps=LMFA01040015 | ||
|SysName=13-Hydroperoxy-9,11,15-Octadecatrienoic Acid/13-Hydroperoxy-9,11,15-Octadecatrienoate | |SysName=13-Hydroperoxy-9,11,15-Octadecatrienoic Acid/13-Hydroperoxy-9,11,15-Octadecatrienoate | ||
|Mass Spectra=EI-MS(Me-ester; after reduction and hydrogenation) | |Mass Spectra=EI-MS(Me-ester; after reduction and hydrogenation)[[Reference:Chan_HWS:,J. Am. Oil Chem. Soc.,1977,54,100|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:,J. Am. Oil Chem. Soc.,1977,54,100}}]][[Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837}}]]: m/e=243[O=CH(CH2)11C(=OH)OCH3]; 214[CH2(CH2)10C(=OH)OCH3]; 211[O=CH(CH2)11C=O], GC-EI-MS(Me-ester; after reduction and TMS)[[Reference:Terao_J:Matsushita_S:,J. Am. Oil Chem. Soc.,1977,54,234|{{RelationTable/GetFirstAuthor|Reference:Terao_J:Matsushita_S:,J. Am. Oil Chem. Soc.,1977,54,234}}]][[Reference:Frankel_EN:Neff_WE:Rohwedder_WK:Khambay_BPS:Garwood_RF:Weedon_BCL:,Lipids,1977,12,1055|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:Neff_WE:Rohwedder_WK:Khambay_BPS:Garwood_RF:Weedon_BCL:,Lipids,1977,12,1055}}]]: m/e=380[M]; 365[M-CH3]; 311[SMTO=CH-CH=CH-CH=CH-(CH2)7-COOCH3] | ||
|UV Spectra=(Me-ester; after reduction; in etoh) | |UV Spectra=(Me-ester; after reduction; in etoh)[[Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837}}]], cis, trans, cis isomer: <FONT FACE="Symbol">l</FONT>max=233nm, trans, trans, cis isomer: <FONT FACE="Symbol">l</FONT>max=232nm | ||
|IR Spectra=(Me-ester; after reduction) | |IR Spectra=(Me-ester; after reduction)[[Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837}}]][[Reference:Frankel_EN:Neff_WE:Rohwedder_WK:Khambay_BPS:Garwood_RF:Weedon_BCL:,Lipids,1977,12,1055|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:Neff_WE:Rohwedder_WK:Khambay_BPS:Garwood_RF:Weedon_BCL:,Lipids,1977,12,1055}}]], cis, trans, cis isomer: 989-983 and 950-945cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>; trans, trans, cis isomer: 992-983cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>, (Me-ester)[[Reference:Toyoda_I:Terao_J:Matsushita_S:,Lipids,1982,17,84|{{RelationTable/GetFirstAuthor|Reference:Toyoda_I:Terao_J:Matsushita_S:,Lipids,1982,17,84}}]], OOH group: 3400cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP> | ||
|NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(cis,trans,cis-isomer) | |NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(cis,trans,cis-isomer)[[Reference:Gardner_HW:Weisleder_D:,Lipids,1972,7,191|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:,Lipids,1972,7,191}}]]: C10: 5.95ppm; C11: 6.54ppm; C12: 5.54ppm; C13: 4.38ppm; J10-11=11Hz; J11-12=15Hz[C11-12: trans]; J12-13=8Hz <SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(cis,trans,cis-isomer; after reduction)[[Reference:Gardner_HW:Weisleder_D:,Lipids,1972,7,191|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:,Lipids,1972,7,191}}]]: C10: 5.94ppm; C11: 6.49ppm; C12: 5.64ppm; C13: 4.20ppm | ||
}} | }} | ||
Revision as of 00:00, 12 December 2008
| IDs and Links | |
|---|---|
| LipidBank | DFA8052 |
| LipidMaps | LMFA01040015 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18303HP03 |
| GlcNAca/b1-3Xyla-4Galb1-3GalNAca1-4(NeuAc?1-2NeuGc4Mea1-3)GalNAcb1-4(EtnP-6)GlcNAcb1-3Manb1-4Glcb1-1Cer | |
|---|---|
| |
| Structural Information | |
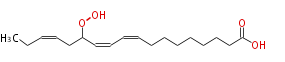
| 13-Hydroperoxy-9,11,15-Octadecatrienoic Acid/13-Hydroperoxy-9,11,15-Octadecatrienoate | |
| Formula | C18H30O4 |
| Exact Mass | 310.21440944799997 |
| Average Mass | 310.4284 |
| SMILES | CCC=CCC(OO)C=CC=CCCCCCCCC(O)=O |
| Physicochemical Information | |
| Spectral Information | |
| Mass Spectra | EI-MS(Me-ester; after reduction and hydrogenation) Chan_HWS Chan_HWS et al.: m/e=243[O=CH(CH2)11C(=OH)OCH3]; 214[CH2(CH2)10C(=OH)OCH3]; 211[O=CH(CH2)11C=O], GC-EI-MS(Me-ester; after reduction and TMS) TeraoJet al. Frankel_EN et al.: m/e=380[M]; 365[M-CH3]; 311[SMTO=CH-CH=CH-CH=CH-(CH2)7-COOCH3] |
| UV Spectra | (Me-ester; after reduction; in etoh) Chan_HWS et al., cis, trans, cis isomer: lmax=233nm, trans, trans, cis isomer: lmax=232nm |
| IR Spectra | (Me-ester; after reduction) Chan_HWS et al. Frankel_EN et al., cis, trans, cis isomer: 989-983 and 950-945cm-1; trans, trans, cis isomer: 992-983cm-1, (Me-ester) ToyodaIet al., OOH group: 3400cm-1 |
| NMR Spectra | 1H-NMR(cis,trans,cis-isomer) Gardner_HW et al.: C10: 5.95ppm; C11: 6.54ppm; C12: 5.54ppm; C13: 4.38ppm; J10-11=11Hz; J11-12=15Hz[C11-12: trans]; J12-13=8Hz 1H-NMR(cis,trans,cis-isomer; after reduction) Gardner_HW et al.: C10: 5.94ppm; C11: 6.49ppm; C12: 5.64ppm; C13: 4.20ppm |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|