LBF20406CV03: Difference between revisions
No edit summary |
No edit summary |
||
| (9 intermediate revisions by the same user not shown) | |||
| Line 6: | Line 6: | ||
|LipidBank=XPR8042 | |LipidBank=XPR8042 | ||
|LipidMaps=LMFA03120023 | |LipidMaps=LMFA03120023 | ||
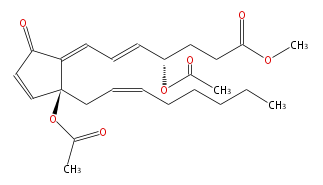
|SysName= | |SysName=Methyl-4R- (trans-5,trans-7) -4-acetoxy-7- [2R-acetoxy-2- (cis-2-octenyl) -5-oxo-3-cyclopentenylidene] -5-heptenoate | ||
|Common Name=&&4- | |Common Name=&&4-epi Clavulone II&&Methyl-4R- (5E,7E) -4-acetoxy-7- [2R-acetoxy-2- (2Z-octenyl) -5-oxo-3-cyclopentenylidene] -5-heptenoate&& | ||
|Optical=[alpha]_D -18.7°(C 0.30, CHCl_3 )[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | |Optical=[ alpha ]_D -18.7°(C 0.30, CHCl_3 )[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | ||
|Mass Spectra=EIMS m/z 446 (M^+ ). HREIMS m/z 446.2315 for | |Mass Spectra=EIMS m/z 446 (M^+ ). HREIMS m/z 446.2315 for C_{25}H_{34}O_7 (M^+ ), calcd 446.2305.[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | ||
|UV Spectra=lambda^ | |UV Spectra= lambda ^{EtOH}_{max} 228 nm(log epsilon 4.28),291 nm(log epsilon 4.27)[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | ||
|IR Spectra=nu^ | |IR Spectra= nu ^{film}_{max}1738,1732,1704,1644,and 1232cm^{-1}[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | ||
|NMR Spectra=^1 H-NMR(500MHz,CDCl_3 ) | |NMR Spectra=^1 H-NMR(500MHz,CDCl_3 ) delta ppm0.88(3H,t,J=7.2Hz),1.20-1.34(6H,m),1.94(2H,q,J=6.8Hz),1.98-2.06(2H,m),2.03(3H,s),2.09(3H,s),2.37(2H,t,J=7.4Hz),2.71(1H,dd,J=8.4,14.1Hz),2.98(1H,dd,J=7.0,14.1Hz),3.67(3H,s),5.18(1H,ddd,J=7.0,8.4,10.9Hz),5.43(1H,dt,J=6.4,7.0Hz),5.50(1H,dt,J=7.4,10.9Hz),6.07(1H,dd,J=6.4,11.9Hz),6.41(1H,d,J=6.1Hz),6.70(1H,ddd,J=1.0,11.9,15.1Hz),6.88(1H,d,J=15.1Hz),7.48(1H,d,J=6.1Hz).[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] ^{13}C-NMR(125MHz,CDCl_3 ) delta ppm14.0,20.9,21.2,22.5,27.4,29.1,29.2,29.6,31.5,35.9,51.8,72.7,85.3,121.1,126.0,129.4,135.1,135.1,136.7,141.5,157.9,169.2,169.9,172.9,193.4.[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | ||
|Source=4-Epiclavulones were isolated from Japanese soft coral, Stolonifer Clavularia viridis Quoy and Gaimard.[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | |Source=4-Epiclavulones were isolated from Japanese soft coral, Stolonifer Clavularia viridis Quoy and Gaimard.[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | ||
|Chemical Synthesis=4-Epiclavulone II was synthesized from clavulone II.[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | |Chemical Synthesis=4-Epiclavulone II was synthesized from clavulone II.[[Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Miyai_Y:Iguchi_K:,Chem. Pharm. Bull. (Tokyo),1999,47,884}}]] | ||
Latest revision as of 05:49, 21 October 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR8042 |
| LipidMaps | LMFA03120023 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20406CV03 |
| 4-epi Clavulone II | |
|---|---|
| |
| Structural Information | |
| Methyl-4R- (trans-5,trans-7) -4-acetoxy-7- [2R-acetoxy-2- (cis-2-octenyl) -5-oxo-3-cyclopentenylidene] -5-heptenoate | |
| |
| Formula | C25H34O7 |
| Exact Mass | 446.230453442 |
| Average Mass | 446.53326000000004 |
| SMILES | O(C(C)=O)[C@@](C1=CC=C[C@H](CCC(OC)=O)OC(C)=O)(CC=CCCCCC)C=CC1=O |
| Physicochemical Information | |
| [ α ]D -18.7°(C 0.30, CHCl3) IwashimaMet al. | |
| 4-Epiclavulones were isolated from Japanese soft coral, Stolonifer Clavularia viridis Quoy and Gaimard. Iwashima_M et al. | |
| 4-Epiclavulone II was synthesized from clavulone II. Iwashima_M et al. | |
| Spectral Information | |
| Mass Spectra | EIMS m/z 446 (M+). HREIMS m/z 446.2315 for C25}H_{34O7 (M+), calcd 446.2305. IwashimaMet al. |
| UV Spectra | λ EtOH max 228 nm(log ε 4.28),291 nm(log ε 4.27) IwashimaMet al. |
| IR Spectra | ν film max 1738,1732,1704,1644,and 1232cm-1 IwashimaMet al. |
| NMR Spectra | 1H-NMR(500MHz,CDCl3) δ ppm0.88(3H,t,J=7.2Hz),1.20-1.34(6H,m),1.94(2H,q,J=6.8Hz),1.98-2.06(2H,m),2.03(3H,s),2.09(3H,s),2.37(2H,t,J=7.4Hz),2.71(1H,dd,J=8.4,14.1Hz),2.98(1H,dd,J=7.0,14.1Hz),3.67(3H,s),5.18(1H,ddd,J=7.0,8.4,10.9Hz),5.43(1H,dt,J=6.4,7.0Hz),5.50(1H,dt,J=7.4,10.9Hz),6.07(1H,dd,J=6.4,11.9Hz),6.41(1H,d,J=6.1Hz),6.70(1H,ddd,J=1.0,11.9,15.1Hz),6.88(1H,d,J=15.1Hz),7.48(1H,d,J=6.1Hz). IwashimaMet al. 13C-NMR(125MHz,CDCl3) δ ppm14.0,20.9,21.2,22.5,27.4,29.1,29.2,29.6,31.5,35.9,51.8,72.7,85.3,121.1,126.0,129.4,135.1,135.1,136.7,141.5,157.9,169.2,169.9,172.9,193.4. IwashimaMet al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|