LBF10000BC02: Difference between revisions
No edit summary |
No edit summary |
||
| Line 12: | Line 12: | ||
|Optical=<FONT FACE="Symbol">h</FONT> 24/D = 1.4432 [[Reference:Lederer_E:Portelance_V:Serck-Hanssen_K:,Bull. Soc. Chim. Fr.,1952,,413|{{RelationTable/GetFirstAuthor|Reference:Lederer_E:Portelance_V:Serck-Hanssen_K:,Bull. Soc. Chim. Fr.,1952,,413}}]] | |Optical=<FONT FACE="Symbol">h</FONT> 24/D = 1.4432 [[Reference:Lederer_E:Portelance_V:Serck-Hanssen_K:,Bull. Soc. Chim. Fr.,1952,,413|{{RelationTable/GetFirstAuthor|Reference:Lederer_E:Portelance_V:Serck-Hanssen_K:,Bull. Soc. Chim. Fr.,1952,,413}}]] | ||
|Source= | |Source= | ||
|Chemical Synthesis=It was synthesized from 2-hexyl-2-decenoic acid by hydrogenation with Pt in ethanol[[Reference:Lederer_E:Portelance_V:Serck-Hanssen_K:,Bull. Soc. Chim. Fr.,1952,,413|{{RelationTable/GetFirstAuthor|Reference:Lederer_E:Portelance_V:Serck-Hanssen_K:,Bull. Soc. Chim. Fr.,1952,,413}}]] | |Chemical Synthesis=It was synthesized from 2-hexyl-2-decenoic acid by hydrogenation with Pt in ethanol[[Reference:Lederer_E:Portelance_V:Serck-Hanssen_K:,Bull. Soc. Chim. Fr.,1952,,413|{{RelationTable/GetFirstAuthor|Reference:Lederer_E:Portelance_V:Serck-Hanssen_K:,Bull. Soc. Chim. Fr.,1952,,413}}]]. | ||
|Metabolism= | |Metabolism= | ||
}} | }} | ||
{{Lipid/Footer}} | {{Lipid/Footer}} | ||
Revision as of 11:55, 25 November 2009
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA7134 |
| LipidMaps | LMFA01020166 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF10000BC02 |
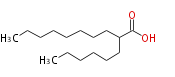
| 2-Hexyldecanoic Acid | |
|---|---|
| |
| Structural Information | |
| 2-Hexyldecanoic Acid | |
| |
| Formula | C16H32O2 |
| Exact Mass | 256.240230268 |
| Average Mass | 256.42408 |
| SMILES | CCCCCCCCC(CCCCCC)C(O)=O |
| Physicochemical Information | |
| 140-150°C/0.02mmHg Lederer_E et al. | |
| D70/4 = 0.8463 MetzgerAet al. | |
| h 24/D = 1.4432 LedererEet al. | |
| It was synthesized from 2-hexyl-2-decenoic acid by hydrogenation with Pt in ethanol Lederer_E et al.. | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|