LBF20307PG38: Difference between revisions
No edit summary |
No edit summary |
||
| Line 14: | Line 14: | ||
|NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(CDCl<SUB><FONT SIZE=-1>3</FONT></SUB>) : <FONT FACE="Symbol">d</FONT> 7.75-7.55(m, 1H, 9-CH), 6.30-6.10(m, 1H, 10-CH), 5.90(brs, 2H, OH), 5.75-5.35(m, 4H), 4.30-3.95(m, 1H, 15-CH) [[Reference:Bundy_GL:Morton_DR:Peterson_DC:Nishizawa_EE:Miller_WL:,J. Med. Chem.,1983,26,790|{{RelationTable/GetFirstAuthor|Reference:Bundy_GL:Morton_DR:Peterson_DC:Nishizawa_EE:Miller_WL:,J. Med. Chem.,1983,26,790}}]] | |NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(CDCl<SUB><FONT SIZE=-1>3</FONT></SUB>) : <FONT FACE="Symbol">d</FONT> 7.75-7.55(m, 1H, 9-CH), 6.30-6.10(m, 1H, 10-CH), 5.90(brs, 2H, OH), 5.75-5.35(m, 4H), 4.30-3.95(m, 1H, 15-CH) [[Reference:Bundy_GL:Morton_DR:Peterson_DC:Nishizawa_EE:Miller_WL:,J. Med. Chem.,1983,26,790|{{RelationTable/GetFirstAuthor|Reference:Bundy_GL:Morton_DR:Peterson_DC:Nishizawa_EE:Miller_WL:,J. Med. Chem.,1983,26,790}}]] | ||
|Source= | |Source= | ||
|Chemical Synthesis=[[Reference:Bundy_GL:Morton_DR:Peterson_DC:Nishizawa_EE:Miller_WL:,J. Med. Chem.,1983,26,790|{{RelationTable/GetFirstAuthor|Reference:Bundy_GL:Morton_DR:Peterson_DC:Nishizawa_EE:Miller_WL:,J. Med. Chem.,1983,26,790}}]];> {{Image200| | |Chemical Synthesis=[[Reference:Bundy_GL:Morton_DR:Peterson_DC:Nishizawa_EE:Miller_WL:,J. Med. Chem.,1983,26,790|{{RelationTable/GetFirstAuthor|Reference:Bundy_GL:Morton_DR:Peterson_DC:Nishizawa_EE:Miller_WL:,J. Med. Chem.,1983,26,790}}]];> {{Image200|LBF20307PG38FT0001.gif}} | ||
|Metabolism=In aqueous solution prostaglandin D2 undergoes non-enzymatic dehydration and is converted to prostaglandin J2 [[Reference:Fukushima_M:,Prostaglandins Leukot. Essent. Fatty Acids,1992,47,1|{{RelationTable/GetFirstAuthor|Reference:Fukushima_M:,Prostaglandins Leukot. Essent. Fatty Acids,1992,47,1}}]];>. | |Metabolism=In aqueous solution prostaglandin D2 undergoes non-enzymatic dehydration and is converted to prostaglandin J2 [[Reference:Fukushima_M:,Prostaglandins Leukot. Essent. Fatty Acids,1992,47,1|{{RelationTable/GetFirstAuthor|Reference:Fukushima_M:,Prostaglandins Leukot. Essent. Fatty Acids,1992,47,1}}]];>. | ||
}} | }} | ||
{{Lipid/Footer}} | {{Lipid/Footer}} | ||
Revision as of 10:00, 25 November 2009
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR1901 |
| LipidMaps | LMFA03010019 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20307PG38 |
| PROSTAGLANDIN J2 | |
|---|---|
| |
| Structural Information | |
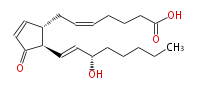
| 7- [ 2 (R) - (3 (S) -Hydroxy-1 (E) -octenyl) -3-oxo-4-cyclopenten-1 (R) -yl ] -5 (Z) -heptenoic acid | |
| |
| Formula | C20H30O4 |
| Exact Mass | 334.21440944799997 |
| Average Mass | 334.4498 |
| SMILES | C(CC[C@@H](O)C=C[C@@H](C(=O)1)[C@@H](CC=CCCCC(O)=O)C=C1)CC |
| Physicochemical Information | |
| ACETONITRILECHLOROFORM, ETHANOL Bundy_GL et al. | |
Bundy_GL et al.;>  | |
| In aqueous solution prostaglandin D2 undergoes non-enzymatic dehydration and is converted to prostaglandin J2 Fukushima_M ;>. | |
| Spectral Information | |
| Mass Spectra | TMS ETHER ; M+ 478.2934 Bundy_GL et al. |
| UV Spectra | l MeOHmax = 305(e 1200), 216(e 9900)nm Bundy_GL et al. |
| IR Spectra | n 3400, 3200, 2660, 1710, 1085, 970 cm-1 Bundy_GL et al. |
| NMR Spectra | 1H-NMR(CDCl3) : d 7.75-7.55(m, 1H, 9-CH), 6.30-6.10(m, 1H, 10-CH), 5.90(brs, 2H, OH), 5.75-5.35(m, 4H), 4.30-3.95(m, 1H, 15-CH) Bundy_GL et al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|