LBF11102SC01: Difference between revisions
No edit summary |
No edit summary |
||
| (6 intermediate revisions by 2 users not shown) | |||
| Line 7: | Line 7: | ||
|LipidMaps=LMFA01030035 | |LipidMaps=LMFA01030035 | ||
|SysName=9-Undecenoic acid | |SysName=9-Undecenoic acid | ||
|Common Name=&&9-Hendecenoic acid&&9- | |Common Name=&&9-Hendecenoic acid&&9-Undecylenic acid&& | ||
|Melting Point=11.4°C | |Melting Point=11.4°C | ||
|Boiling Point=129°C at 1 mmHg | |Boiling Point=129°C at 1 mmHg | ||
|Solubility=[[Reference:Ahmad_K:Bumpus_FM:Strong_FM:,J. Am. Chem. Soc.,1948,70,3391|{{RelationTable/GetFirstAuthor|Reference:Ahmad_K:Bumpus_FM:Strong_FM:,J. Am. Chem. Soc.,1948,70,3391}}]] | |Solubility=[[Reference:Ahmad_K:Bumpus_FM:Strong_FM:,J. Am. Chem. Soc.,1948,70,3391|{{RelationTable/GetFirstAuthor|Reference:Ahmad_K:Bumpus_FM:Strong_FM:,J. Am. Chem. Soc.,1948,70,3391}}]]<!--0017--><!--0069--> | ||
|Source= | |Source= | ||
|Chemical Synthesis=Synthetic by debromination of 9-bromo- | |Chemical Synthesis=Synthetic by debromination of 9-bromo-9-undecenoic acid with (i)Na in ethanol,or (ii)Zn and propionic acid. | ||
|Metabolism= | |Metabolism= | ||
|Symbol=C11:1 | |Symbol=C11:1 | ||
Latest revision as of 10:44, 6 September 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA0074 |
| LipidMaps | LMFA01030035 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF11102SC01 |
| 9-Hendecenoic acid | |
|---|---|
| |
| Structural Information | |
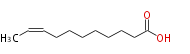
| 9-Undecenoic acid | |
| |
| C11:1 | |
| Formula | C11H20O2 |
| Exact Mass | 184.14632988399998 |
| Average Mass | 184.2753 |
| SMILES | CC=CCCCCCCCC(O)=O |
| Physicochemical Information | |
| 11.4°C | |
| 129°C at 1 mmHg | |
| AhmadKet al. | |
| Synthetic by debromination of 9-bromo-9-undecenoic acid with (i)Na in ethanol,or (ii)Zn and propionic acid. | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|