LBF18108HP01: Difference between revisions
No edit summary |
No edit summary |
||
| (2 intermediate revisions by the same user not shown) | |||
| Line 11: | Line 11: | ||
|IR Spectra=Isorated trans unsaturation(970cm^{-1}), trans epoxide(885cm^{-1}), OOH(3600 AND 3430cm^{-1}) [[Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246}}]] | |IR Spectra=Isorated trans unsaturation(970cm^{-1}), trans epoxide(885cm^{-1}), OOH(3600 AND 3430cm^{-1}) [[Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246}}]] | ||
|NMR Spectra=^1 H-NMR(methyl ester)[[Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246}}]]: C9(4.33ppm), C10(5.85ppm), C11(5.47ppm), C12(3.11ppm), C13(2.84ppm) J10-11=16Hz(trans olefin), J12-13=2Hz(trans epoxide) | |NMR Spectra=^1 H-NMR(methyl ester)[[Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246}}]]: C9(4.33ppm), C10(5.85ppm), C11(5.47ppm), C12(3.11ppm), C13(2.84ppm) J10-11=16Hz(trans olefin), J12-13=2Hz(trans epoxide) | ||
|Source=Major reactive products between 13-hydroperoxylinoleate (or linoleate) and soy bean extracts(pH=6.9)[[Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:Kleiman_R | |Source=Major reactive products between 13-hydroperoxylinoleate (or linoleate) and soy bean extracts(pH=6.9)[[Reference:Gardner_HW:Weisleder_D:Kleiman_R:,Lipids,1978,13,246|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:Kleiman_R,Lipids,1978,13,246}}]]. Copper-catalyzed degradation products of linoleate methylhydroperoxide[[Reference:Wu_GS:Stein_RA:Mead_JF:,Lipids,1977,12,971|{{RelationTable/GetFirstAuthor|Reference:Wu_GS:Stein_RA:Mead_JF:,Lipids,1977,12,971}}]]. Production mechanism [[Reference:Frankel_EN:,Prog. Lipid Res.,1984,23,197|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:,Prog. Lipid Res.,1984,23,197}}]]<!--8030-->[[Reference:Gardner_HW:,J. Agric. Food Chem.,1975,23,129|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:,J. Agric. Food Chem.,1975,23,129}}]]<!--8074-->. | ||
|Chemical Synthesis= | |Chemical Synthesis= | ||
|Metabolism= | |Metabolism= | ||
Latest revision as of 17:31, 4 November 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA8009 |
| LipidMaps | LMFA01040010 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18108HP01 |
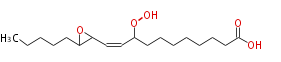
| 12,13-Epoxy-9-hydroperoxy-10-octadecenoic acid | |
|---|---|
| |
| Structural Information | |
| 12,13-Epoxy-9-hydroperoxy-10-octadecenoic acid | |
| |
| Formula | C18H32O5 |
| Exact Mass | 328.224974134 |
| Average Mass | 328.44368000000003 |
| SMILES | C(C(C=CC(OO)CCCCCCCC(O)=O)1)(CCCCC)O1 |
| Physicochemical Information | |
| Major reactive products between 13-hydroperoxylinoleate (or linoleate) and soy bean extracts(pH=6.9) Gardner_HW et al.. Copper-catalyzed degradation products of linoleate methylhydroperoxide Wu_GS et al.. Production mechanism Frankel_EN Gardner_HW . | |
| It showed a slightly higher toxicity than linoleate monohydroxyperoxide. Fujimoto_K | |
| Spectral Information | |
| Mass Spectra | GC-EI-MS(after methanolysis, reduction and trimethylsilylation) Gardner_HW et al. TokitaMet al. |
| UV Spectra | |
| IR Spectra | Isorated trans unsaturation(970cm-1), trans epoxide(885cm-1), OOH(3600 AND 3430cm-1) Gardner_HW et al. |
| NMR Spectra | 1H-NMR(methyl ester) Gardner_HW et al.: C9(4.33ppm), C10(5.85ppm), C11(5.47ppm), C12(3.11ppm), C13(2.84ppm) J10-11=16Hz(trans olefin), J12-13=2Hz(trans epoxide) |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|