LBF20406AM39: Difference between revisions
No edit summary |
No edit summary |
||
| (17 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
{{Lipid/Header}} | |||
{{Hierarchy|{{PAGENAME}}}} | {{Hierarchy|{{PAGENAME}}}} | ||
| Line 4: | Line 6: | ||
|LipidBank=XPR7061 | |LipidBank=XPR7061 | ||
|LipidMaps=LMFA08020047 | |LipidMaps=LMFA08020047 | ||
|SysName=N- ( (R) - (-) -2- | |SysName=N-l-2R-Hydroxypropyl-alpha,alpha-dimethyl- (cis-5,cis-8,cis-11,cis-14) -eicosatetraenoylamine | ||
|Common Name=&&N- (R) - (-) -2-Hydroxypropyl-N- (R) - (-) -2-Hydroxypropyl-alpha,alpha-dimethylachidonoylamide&&N- (R) - (-) -2-Hydroxypropyl-alpha,alpha-dimethyl- (5Z,8Z,11Z,14Z) -eicosatetraenoylamine&& | |||
|Melting Point=colorless oil [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | |Melting Point=colorless oil [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | ||
|NMR Spectra= | |NMR Spectra=^1 H NMR (CDCl3) delta 5.95 (br s, l H), 5.30-5.42 (m, 8H), 3.92-3.96 (m, 1H), 3.40-3.50 (m, 1H), 3.02-3.23 (m, 1H), 2.70-2.95 (m, 7H), 2.20-2.28(m, 1H), 2.00-2.15 (m, 4H), 1.70-1.75 (m, 2H), 1.20-1.52 (series of m, 14H), 0.89 (t, J=6.7 Hz, 3H), [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | ||
|Source= | |||
|Chemical Synthesis=This compound was synthesized from arachidonic acid and (R)-(-)-l-amino-2-propanolyield 63%. [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | |||
|Metabolism= | |||
|Biological Activity=Binding to the brain cannabinoid receptor(CBl), Ki(nM)= 153.9±30.0 [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | |||
}} | }} | ||
{{Lipid/Footer}} | |||
Latest revision as of 13:49, 21 October 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR7061 |
| LipidMaps | LMFA08020047 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20406AM39 |
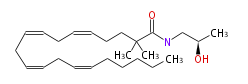
| N- (R) - (-) -2-Hydroxypropyl-N- (R) - (-) -2-Hydroxypropyl-α,α-dimethylachidonoylamide | |
|---|---|
| |
| Structural Information | |
| N-l-2R-Hydroxypropyl-α,α-dimethyl- (cis-5,cis-8,cis-11,cis-14) -eicosatetraenoylamine | |
| |
| Formula | C25H43NO2 |
| Exact Mass | 389.329379625 |
| Average Mass | 389.61446 |
| SMILES | CC(C)(C(=O)NC[C@@H](C)O)CCC=CCC=CCC=CCC=CCCCCC |
| Physicochemical Information | |
| colorless oil Sheskin_T et al. | |
| This compound was synthesized from arachidonic acid and (R)-(-)-l-amino-2-propanolyield 63%. Sheskin_T et al. | |
| Binding to the brain cannabinoid receptor(CBl), Ki(nM)= 153.9±30.0 Sheskin_T et al. | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | 1H NMR (CDCl3) δ 5.95 (br s, l H), 5.30-5.42 (m, 8H), 3.92-3.96 (m, 1H), 3.40-3.50 (m, 1H), 3.02-3.23 (m, 1H), 2.70-2.95 (m, 7H), 2.20-2.28(m, 1H), 2.00-2.15 (m, 4H), 1.70-1.75 (m, 2H), 1.20-1.52 (series of m, 14H), 0.89 (t, J=6.7 Hz, 3H), SheskinTet al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|