LBF18303HP03: Difference between revisions
No edit summary |
No edit summary |
||
| (18 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
{{Lipid/Header}} | |||
{{Hierarchy|{{PAGENAME}}}} | {{Hierarchy|{{PAGENAME}}}} | ||
| Line 4: | Line 6: | ||
|LipidBank=DFA8052 | |LipidBank=DFA8052 | ||
|LipidMaps=LMFA01040015 | |LipidMaps=LMFA01040015 | ||
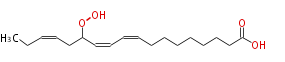
|SysName=13-Hydroperoxy-9,11,15- | |SysName=13-Hydroperoxy-9,11,15-octadecatrienoic acid | ||
|Mass Spectra=EI-MS(Me-ester; after reduction and hydrogenation) | |Common Name=&&13-HpOTrE&& | ||
|UV Spectra=(Me-ester; after reduction; in | |Mass Spectra=EI-MS(Me-ester; after reduction and hydrogenation)[[Reference:Chan_HWS:,J. Am. Oil Chem. Soc.,1977,54,100|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:,J. Am. Oil Chem. Soc.,1977,54,100}}]][[Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837}}]]: m/e=243[O=CH(CH2)11C(=OH)OCH3]; 214[CH2(CH2)10C(=OH)OCH3]; 211[O=CH(CH2)11C=O], GC-EI-MS(Me-ester; after reduction and TMS)[[Reference:Terao_J:Matsushita_S:,J. Am. Oil Chem. Soc.,1977,54,234|{{RelationTable/GetFirstAuthor|Reference:Terao_J:Matsushita_S:,J. Am. Oil Chem. Soc.,1977,54,234}}]][[Reference:Frankel_EN:Neff_WE:Rohwedder_WK:Khambay_BPS:Garwood_RF:Weedon_BCL:,Lipids,1977,12,1055|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:Neff_WE:Rohwedder_WK:Khambay_BPS:Garwood_RF:Weedon_BCL:,Lipids,1977,12,1055}}]]: m/e=380[M]; 365[M-CH3]; 311[SMTO=CH-CH=CH-CH=CH-(CH2)7-COOCH3] | ||
|IR Spectra=(Me-ester; after reduction) | |UV Spectra=(Me-ester; after reduction; in ethanol)[[Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837}}]], cis, trans, cis isomer: lambda max=233nm, trans, trans, cis isomer: lambda max=232nm | ||
|NMR Spectra= | |IR Spectra=(Me-ester; after reduction)[[Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:Levett_G:,Lipids,1977,12,837}}]][[Reference:Frankel_EN:Neff_WE:Rohwedder_WK:Khambay_BPS:Garwood_RF:Weedon_BCL:,Lipids,1977,12,1055|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:Neff_WE:Rohwedder_WK:Khambay_BPS:Garwood_RF:Weedon_BCL:,Lipids,1977,12,1055}}]], cis, trans, cis isomer: 989-983 and 950-945cm^{-1}; trans, trans, cis isomer: 992-983cm^{-1}, (Me-ester)[[Reference:Toyoda_I:Terao_J:Matsushita_S:,Lipids,1982,17,84|{{RelationTable/GetFirstAuthor|Reference:Toyoda_I:Terao_J:Matsushita_S:,Lipids,1982,17,84}}]], OOH group: 3400cm^{-1} | ||
|NMR Spectra=^1 H-NMR(cis,trans,cis-isomer)[[Reference:Gardner_HW:Weisleder_D:,Lipids,1972,7,191|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:,Lipids,1972,7,191}}]]: C10: 5.95ppm; C11: 6.54ppm; C12: 5.54ppm; C13: 4.38ppm; J10-11=11Hz; J11-12=15Hz[C11-12: trans]; J12-13=8Hz ^1 H-NMR(cis,trans,cis-isomer; after reduction)[[Reference:Gardner_HW:Weisleder_D:,Lipids,1972,7,191|{{RelationTable/GetFirstAuthor|Reference:Gardner_HW:Weisleder_D:,Lipids,1972,7,191}}]]: C10: 5.94ppm; C11: 6.49ppm; C12: 5.64ppm; C13: 4.20ppm | |||
|Source=Auto oxidation of linoleate<!--8029--><!--8022--><!--8023--><!--8021-->[[Reference:Frankel_EN:,Prog. Lipid Res.,1980,19,1|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:,Prog. Lipid Res.,1980,19,1}}]][[Reference:Frankel_EN:,Prog. Lipid Res.,1983,22,1|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:,Prog. Lipid Res.,1983,22,1}}]][[Reference:Frankel_EN:,Prog. Lipid Res.,1984,23,197|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:,Prog. Lipid Res.,1984,23,197}}]]<!--8027--><!--8030-->[[Reference:Chan_HWS:Coxon_DT:Peers_KE:Price_KR:,Food Chemistry,1982,9,21|{{RelationTable/GetFirstAuthor|Reference:Chan_HWS:Coxon_DT:Peers_KE:Price_KR:,Food Chemistry,1982,9,21}}]]. Oxidation of linoleate by singlet oxygen<!--8022--><!--8023-->[[Reference:Frankel_EN:,Prog. Lipid Res.,1980,19,1|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:,Prog. Lipid Res.,1980,19,1}}]][[Reference:Frankel_EN:,Prog. Lipid Res.,1983,22,1|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:,Prog. Lipid Res.,1983,22,1}}]][[Reference:Frankel_EN:,Prog. Lipid Res.,1984,23,197|{{RelationTable/GetFirstAuthor|Reference:Frankel_EN:,Prog. Lipid Res.,1984,23,197}}]]<!--8027--><!--8030-->. Reaction products between linoleate and soy bean lipoxygenase(pH:8.7,25 | |||
|Chemical Synthesis= | |||
|Metabolism= | |||
|Biological Activity=Pysiological damages are induced by these hydroperoxides which are incorporated into bodies or synthesized endogenously.[[Reference:Logani_MK:Davies_RE:,Lipids,1980,15,485|{{RelationTable/GetFirstAuthor|Reference:Logani_MK:Davies_RE:,Lipids,1980,15,485}}]]<!--8044--><!--8045-->[[Reference:Sevanian_A:Hochstein_P:,Annu. Rev. Nutr.,1985,5,365|{{RelationTable/GetFirstAuthor|Reference:Sevanian_A:Hochstein_P:,Annu. Rev. Nutr.,1985,5,365}}]][[Reference:Fujimoto_K:,Fragrance J. (in Japanese),1986,76,21|{{RelationTable/GetFirstAuthor|Reference:Fujimoto_K:,Fragrance J. (in Japanese),1986,76,21}}]]<!--8048--><!--8049-->. It reacts with DNA in the presence of Fe ions and ascorbic acid[[Reference:Fujimoto_K:Neff_WE:Frankel_EN:,Biochim. Biophys. Acta,1984,795,100|{{RelationTable/GetFirstAuthor|Reference:Fujimoto_K:Neff_WE:Frankel_EN:,Biochim. Biophys. Acta,1984,795,100}}]]. | |||
}} | |||
{{MassbankSpectra| | |||
UT000082 | |||
UT000083 | |||
UT000084 | |||
UT000085 | |||
UT000086 | |||
UT000087 | |||
UT000088 | |||
UT000089 | |||
UT000090 | |||
}} | }} | ||
{{Lipid/Footer}} | |||
Latest revision as of 09:00, 17 January 2014
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA8052 |
| LipidMaps | LMFA01040015 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18303HP03 |
| 13-HpOTrE | |
|---|---|
| |
| Structural Information | |
| 13-Hydroperoxy-9,11,15-octadecatrienoic acid | |
| |
| Formula | C18H30O4 |
| Exact Mass | 310.21440944799997 |
| Average Mass | 310.4284 |
| SMILES | CCC=CCC(OO)C=CC=CCCCCCCCC(O)=O |
| Physicochemical Information | |
| Auto oxidation of linoleate Frankel_EN Frankel_EN Frankel_EN Chan_HWS et al.. Oxidation of linoleate by singlet oxygen Frankel_EN Frankel_EN Frankel_EN . Reaction products between linoleate and soy bean lipoxygenase(pH:8.7,25 | |
| Pysiological damages are induced by these hydroperoxides which are incorporated into bodies or synthesized endogenously. Logani_MK et al. Sevanian_A et al. Fujimoto_K . It reacts with DNA in the presence of Fe ions and ascorbic acid Fujimoto_K et al.. | |
| Spectral Information | |
| Mass Spectra | EI-MS(Me-ester; after reduction and hydrogenation) Chan_HWS Chan_HWS et al.: m/e=243[O=CH(CH2)11C(=OH)OCH3]; 214[CH2(CH2)10C(=OH)OCH3]; 211[O=CH(CH2)11C=O], GC-EI-MS(Me-ester; after reduction and TMS) TeraoJet al. Frankel_EN et al.: m/e=380[M]; 365[M-CH3]; 311[SMTO=CH-CH=CH-CH=CH-(CH2)7-COOCH3] |
| UV Spectra | (Me-ester; after reduction; in ethanol) Chan_HWS et al., cis, trans, cis isomer: λ max=233nm, trans, trans, cis isomer: λ max=232nm |
| IR Spectra | (Me-ester; after reduction) Chan_HWS et al. Frankel_EN et al., cis, trans, cis isomer: 989-983 and 950-945cm-1; trans, trans, cis isomer: 992-983cm-1, (Me-ester) ToyodaIet al., OOH group: 3400cm-1 |
| NMR Spectra | 1H-NMR(cis,trans,cis-isomer) Gardner_HW et al.: C10: 5.95ppm; C11: 6.54ppm; C12: 5.54ppm; C13: 4.38ppm; J10-11=11Hz; J11-12=15Hz[C11-12: trans]; J12-13=8Hz 1H-NMR(cis,trans,cis-isomer; after reduction) Gardner_HW et al.: C10: 5.94ppm; C11: 6.49ppm; C12: 5.64ppm; C13: 4.20ppm |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|








