LBF18207HO04: Difference between revisions
No edit summary |
No edit summary |
||
| (7 intermediate revisions by the same user not shown) | |||
| Line 6: | Line 6: | ||
|LipidBank=DFA8029 | |LipidBank=DFA8029 | ||
|LipidMaps=LMFA01050131 | |LipidMaps=LMFA01050131 | ||
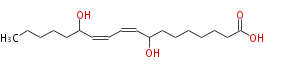
|SysName=8,13-Dihydroxy-9,11- | |SysName=8,13-Dihydroxy-9,11-octadecadienoic acid | ||
|Common Name=&&8,13-Dihydroxy-9,11- | |Common Name=&&8,13-Dihydroxy-9,11-octadecadienoic acid&& | ||
|Mass Spectra=GC-EI-MS(after methanolysis and trimethylsilylation)[[Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191}}]]: m/e=455[M-CH3], 439[M-OCH3], 380[M-HOTMS], 245[SMTO=CH-(CH2)6COOCH3], 237[M-(CH2)6COOCH3-HOTMS], 173[SMTO=CH-(CH2)4CH3], GC-EI-MS(after methanolysis, hydrogenation and trimethylsilylation)[[Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191}}]]: m/e=459[M-CH3], 403[M-(CH2)4CH3], 374[(CH2)4 -CH(OTMS)-(CH2)6-C(OTMS)OCH] | |Mass Spectra=GC-EI-MS(after methanolysis and trimethylsilylation)[[Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191}}]]: m/e=455[M-CH3], 439[M-OCH3], 380[M-HOTMS], 245[SMTO=CH-(CH2)6COOCH3], 237[M-(CH2)6COOCH3-HOTMS], 173[SMTO=CH-(CH2)4CH3], GC-EI-MS(after methanolysis, hydrogenation and trimethylsilylation)[[Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191}}]]: m/e=459[M-CH3], 403[M-(CH2)4CH3], 374[(CH2)4 -CH(OTMS)-(CH2)6-C(OTMS)OCH] | ||
|UV Spectra=Methyl ester: | |UV Spectra=Methyl ester: lambda MeOH/max=230nm(224nm and 238nm)[[Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191}}]][[Reference:Hamberg_M:,Biochim. Biophys. Acta - Lipids and Lipid Metabolism,1983,752,353|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta - Lipids and Lipid Metabolism,1983,752,353}}]] | ||
|IR Spectra=trans trans unsaturation(990-981cm | |IR Spectra=trans trans unsaturation(990-981cm^{-1})[[Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191}}]][[Reference:Hamberg_M:,Biochim. Biophys. Acta - Lipids and Lipid Metabolism,1983,752,353|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta - Lipids and Lipid Metabolism,1983,752,353}}]] | ||
|Source=Reaction products between 13-Hydroperoxylinoleate and human hemoglobin in EtOH[[Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191}}]] | |Source=Reaction products between 13-Hydroperoxylinoleate and human hemoglobin in EtOH[[Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta,1983,752,191}}]]. Autooxydation products of linoleic acid in EtOH-water[[Reference:Hamberg_M:,Biochim. Biophys. Acta - Lipids and Lipid Metabolism,1983,752,353|{{RelationTable/GetFirstAuthor|Reference:Hamberg_M:,Biochim. Biophys. Acta - Lipids and Lipid Metabolism,1983,752,353}}]]. | ||
|Chemical Synthesis= | |Chemical Synthesis= | ||
|Metabolism= | |Metabolism= | ||
Latest revision as of 06:00, 15 April 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA8029 |
| LipidMaps | LMFA01050131 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18207HO04 |
| 8,13-Dihydroxy-9,11-octadecadienoic acid | |
|---|---|
| |
| Structural Information | |
| 8,13-Dihydroxy-9,11-octadecadienoic acid | |
| |
| Formula | C18H32O4 |
| Exact Mass | 312.23005951199997 |
| Average Mass | 312.44428 |
| SMILES | CCCCCC(O)C=CC=CC(O)CCCCCCC(O)=O |
| Physicochemical Information | |
| Reaction products between 13-Hydroperoxylinoleate and human hemoglobin in EtOH Hamberg_M . Autooxydation products of linoleic acid in EtOH-water Hamberg_M . | |
| Spectral Information | |
| Mass Spectra | GC-EI-MS(after methanolysis and trimethylsilylation) HambergM: m/e=455[M-CH3], 439[M-OCH3], 380[M-HOTMS], 245[SMTO=CH-(CH2)6COOCH3], 237[M-(CH2)6COOCH3-HOTMS], 173[SMTO=CH-(CH2)4CH3], GC-EI-MS(after methanolysis, hydrogenation and trimethylsilylation) HambergM: m/e=459[M-CH3], 403[M-(CH2)4CH3], 374[(CH2)4 -CH(OTMS)-(CH2)6-C(OTMS)OCH] |
| UV Spectra | Methyl ester: λ MeOH/max=230nm(224nm and 238nm) HambergM HambergM |
| IR Spectra | trans trans unsaturation(990-981cm-1) HambergM HambergM |
| NMR Spectra | |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|