LBF20307PG03: Difference between revisions
No edit summary |
No edit summary |
||
| (16 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
{{Lipid/Header}} | |||
{{Hierarchy|{{PAGENAME}}}} | {{Hierarchy|{{PAGENAME}}}} | ||
| Line 4: | Line 6: | ||
|LipidBank=XPR1101 | |LipidBank=XPR1101 | ||
|LipidMaps=LMFA03010018 | |LipidMaps=LMFA03010018 | ||
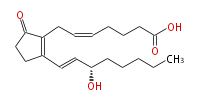
|SysName=7- [ | |SysName=7- [2R- (3S-hydroxy-trans-1-octenyl) -5-oxo-1-cyclopenten-1R-yl] -cis-5-heptenoic acid | ||
|Common Name=&& | |Common Name=&&Prostaglandin B_2&&7- [2R- (3S-Hydroxy-1-(E)-octenyl) -5-oxo-1-cyclopenten-1R-yl] -5-(Z) -heptenoic acid&& | ||
|Mass Spectra=METHYL ESTER; m/e 348(M | |Mass Spectra=METHYL ESTER; m/e 348(M^+ ), 317, 249, 247, 245, 217, 215, 133, 119, 109 [[Reference:Floyd_MB:Schaub_RE:Siuta_GJ:Skotnicki_JS:Grudzinskas_CV:Weiss_MJ:Dessy_F:VanHumbeeck_L:,J. Med. Chem.,1980,23,903|{{RelationTable/GetFirstAuthor|Reference:Floyd_MB:Schaub_RE:Siuta_GJ:Skotnicki_JS:Grudzinskas_CV:Weiss_MJ:Dessy_F:VanHumbeeck_L:,J. Med. Chem.,1980,23,903}}]] | ||
|UV Spectra=MeOH: 278 nm ( | |UV Spectra=MeOH: 278 nm ( epsilon 26000) [[Reference:Floyd_MB:Schaub_RE:Siuta_GJ:Skotnicki_JS:Grudzinskas_CV:Weiss_MJ:Dessy_F:VanHumbeeck_L:,J. Med. Chem.,1980,23,903|{{RelationTable/GetFirstAuthor|Reference:Floyd_MB:Schaub_RE:Siuta_GJ:Skotnicki_JS:Grudzinskas_CV:Weiss_MJ:Dessy_F:VanHumbeeck_L:,J. Med. Chem.,1980,23,903}}]] | ||
|NMR Spectra=METHYL ESTER ; | |NMR Spectra=METHYL ESTER ; ^1 H-NMR : delta 6.9(d, J=16Hz, 1H, 13-CH), 6.3(dd, J=5.5, 16Hz, 1H,14-CH), 5.7-5.1(m, 2H), 4.5-4.1(m, 1H, 15-CH), 3.65(S, 3H, OCH3), 3.15-2.85(m, 2H, 7-CH) [[Reference:Floyd_MB:Schaub_RE:Siuta_GJ:Skotnicki_JS:Grudzinskas_CV:Weiss_MJ:Dessy_F:VanHumbeeck_L:,J. Med. Chem.,1980,23,903|{{RelationTable/GetFirstAuthor|Reference:Floyd_MB:Schaub_RE:Siuta_GJ:Skotnicki_JS:Grudzinskas_CV:Weiss_MJ:Dessy_F:VanHumbeeck_L:,J. Med. Chem.,1980,23,903}}]] | ||
|Source=Prostaglandin B2 was found in human seminal plasma in an amout of 50 micrograms per ml as measured in combination with prostaglandins A1,A2 and B1 [[Reference:Bergstrom_S:,Science,1967,157,382|{{RelationTable/GetFirstAuthor|Reference:Bergstrom_S:,Science,1967,157,382}}]]. | |||
|Chemical Synthesis= | |||
|Metabolism=Prostaglandin E2 is converted to prostaglandin B2 non-enzymatically at alkaline condition [[Reference:Pike_JE:Lincoln_FH:Schneider_WP:,J. Org. Chem.,1969,34,3552|{{RelationTable/GetFirstAuthor|Reference:Pike_JE:Lincoln_FH:Schneider_WP:,J. Org. Chem.,1969,34,3552}}]]. Huma serum contains a dehydrase converting prostaglandin E2 to A2, an isomerase converting prostaglandin A2 to C2, and another isomerase converting prostaglandin C2 to B2 [[Reference:Polet_H:Levine_L:,J. Biol. Chem.,1975,250,351|{{RelationTable/GetFirstAuthor|Reference:Polet_H:Levine_L:,J. Biol. Chem.,1975,250,351}}]]. | |||
|Symbol=PGB2 | |||
}} | }} | ||
{{Lipid/Footer}} | |||
Latest revision as of 18:01, 21 October 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR1101 |
| LipidMaps | LMFA03010018 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20307PG03 |
| Prostaglandin B2 | |
|---|---|
| |
| Structural Information | |
| 7- [2R- (3S-hydroxy-trans-1-octenyl) -5-oxo-1-cyclopenten-1R-yl] -cis-5-heptenoic acid | |
| |
| PGB2 | |
| Formula | C20H30O4 |
| Exact Mass | 334.21440944799997 |
| Average Mass | 334.4498 |
| SMILES | C(CC[C@@H](O)C=CC(=C1CC=CCCCC(O)=O)CCC(=O)1)CC |
| Physicochemical Information | |
| Prostaglandin B2 was found in human seminal plasma in an amout of 50 micrograms per ml as measured in combination with prostaglandins A1,A2 and B1 Bergstrom_S . | |
| Prostaglandin E2 is converted to prostaglandin B2 non-enzymatically at alkaline condition Pike_JE et al.. Huma serum contains a dehydrase converting prostaglandin E2 to A2, an isomerase converting prostaglandin A2 to C2, and another isomerase converting prostaglandin C2 to B2 Polet_H et al.. | |
| Spectral Information | |
| Mass Spectra | METHYL ESTER; m/e 348(M+), 317, 249, 247, 245, 217, 215, 133, 119, 109 Floyd_MB et al. |
| UV Spectra | MeOH: 278 nm ( ε 26000) Floyd_MB et al. |
| IR Spectra | |
| NMR Spectra | METHYL ESTER ; 1H-NMR : δ 6.9(d, J=16Hz, 1H, 13-CH), 6.3(dd, J=5.5, 16Hz, 1H,14-CH), 5.7-5.1(m, 2H), 4.5-4.1(m, 1H, 15-CH), 3.65(S, 3H, OCH3), 3.15-2.85(m, 2H, 7-CH) Floyd_MB et al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|