LBF15306CV01: Difference between revisions
No edit summary |
No edit summary |
||
| Line 8: | Line 8: | ||
|SysName= (Z) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde | |SysName= (Z) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde | ||
|Common Name=&&clavirin I&&(Z) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde&& | |Common Name=&&clavirin I&&(Z) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde&& | ||
| | |Optical=[<FONT FACE="Symbol">a</FONT>]<SUB><FONT SIZE=-1>D</FONT></SUB> -17.1°(C 0.48, CHCl<SUB><FONT SIZE=-1>3</FONT></SUB>) [[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | ||
|Mass Spectra=HREIMS m/z 230.1298 for C<SUB><FONT SIZE=-1>1</FONT></SUB><SUB><FONT SIZE=-1>5</FONT></SUB>H<SUB><FONT SIZE=-1>1</FONT></SUB><SUB><FONT SIZE=-1>8</FONT></SUB>O<SUB><FONT SIZE=-1>2</FONT></SUB> (M<SUP><FONT SIZE=-1>+</FONT></SUP>-CH<SUB><FONT SIZE=-1>3</FONT></SUB>CO<SUB><FONT SIZE=-1>2</FONT></SUB>H), calcd 230.1307. [[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | |Mass Spectra=HREIMS m/z 230.1298 for C<SUB><FONT SIZE=-1>1</FONT></SUB><SUB><FONT SIZE=-1>5</FONT></SUB>H<SUB><FONT SIZE=-1>1</FONT></SUB><SUB><FONT SIZE=-1>8</FONT></SUB>O<SUB><FONT SIZE=-1>2</FONT></SUB> (M<SUP><FONT SIZE=-1>+</FONT></SUP>-CH<SUB><FONT SIZE=-1>3</FONT></SUB>CO<SUB><FONT SIZE=-1>2</FONT></SUB>H), calcd 230.1307. [[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | ||
|IR Spectra=<FONT FACE="Symbol">n</FONT><SUP><FONT SIZE=-1>f</FONT></SUP><SUP><FONT SIZE=-1>i</FONT></SUP><SUP><FONT SIZE=-1>l</FONT></SUP><SUP><FONT SIZE=-1>m</FONT></SUP><SUB><FONT SIZE=-1>m</FONT></SUB><SUB><FONT SIZE=-1>a</FONT></SUB><SUB><FONT SIZE=-1>x</FONT></SUB>1735,1714,1682, and 1227cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP> [[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | |IR Spectra=<FONT FACE="Symbol">n</FONT><SUP><FONT SIZE=-1>f</FONT></SUP><SUP><FONT SIZE=-1>i</FONT></SUP><SUP><FONT SIZE=-1>l</FONT></SUP><SUP><FONT SIZE=-1>m</FONT></SUP><SUB><FONT SIZE=-1>m</FONT></SUB><SUB><FONT SIZE=-1>a</FONT></SUB><SUB><FONT SIZE=-1>x</FONT></SUB>1735,1714,1682, and 1227cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP> [[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | ||
| Line 15: | Line 15: | ||
|Chemical Synthesis=Clavirin I and II were synthesized from 4-alkoxy-2-cyclopentenone.[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | |Chemical Synthesis=Clavirin I and II were synthesized from 4-alkoxy-2-cyclopentenone.[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | ||
|Metabolism=Clavirins may possibly be biosynthesized from clavulone.[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | |Metabolism=Clavirins may possibly be biosynthesized from clavulone.[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | ||
|Biological Activity=Clavirins showed growth-inhibitory activity toward Hela S3 at 1 <FONT FACE="Symbol">m</FONT>g/ml.[[Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455|{{RelationTable/GetFirstAuthor|Reference:Iwashima_M:Okamoto_K:Iguchi_K:,Tetrahedron Lett.,1999,40,6455}}]] | |||
}} | }} | ||
{{Lipid/Footer}} | {{Lipid/Footer}} | ||
Revision as of 06:00, 7 January 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR8044 |
| LipidMaps | LMFA03120025 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF15306CV01 |
| clavirin I | |
|---|---|
| |
| Structural Information | |
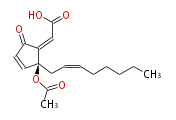
| (Z) -2- [ (R) -2-acetoxy-2- [ (Z) -2-octenyl ] -5-oxo-3-cyclopentenylidene ] acetaldehyde | |
| |
| Formula | C17H22O4 |
| Exact Mass | 290.151809192 |
| Average Mass | 290.35418 |
| SMILES | CCCCCC=CC[C@](OC(C)=O)(C=1)C(=CC=O)C(=O)C1 |
| Physicochemical Information | |
| [a]D -17.1°(C 0.48, CHCl3) IwashimaMet al. | |
| Clavirins were isolated from Japanese soft coral, Stolonifer Clavularia viridis Quoy and Gaimard. Iwashima_M et al. | |
| Clavirin I and II were synthesized from 4-alkoxy-2-cyclopentenone. Iwashima_M et al. | |
| Clavirins may possibly be biosynthesized from clavulone. Iwashima_M et al. | |
| Clavirins showed growth-inhibitory activity toward Hela S3 at 1 mg/ml. Iwashima_M et al. | |
| Spectral Information | |
| Mass Spectra | HREIMS m/z 230.1298 for C15H18O2 (M+-CH3CO2H), calcd 230.1307. IwashimaMet al. |
| UV Spectra | |
| IR Spectra | nfilmmax1735,1714,1682, and 1227cm-1 IwashimaMet al. |
| NMR Spectra | 1H-NMR(500MHz,CDCl3)dppm0.88(3H,t,J=7.1Hz),1.20-1.36(6H,m),1.96(2H,brq,J=7.1Hz),2.05(3H,s),2.68(1H,dd,J=7.7,14.6Hz),2.77(1H,dd,J=7.4,14.6Hz),5.23(1H,dt,J=7.1,10.9Hz),5.59(1H,ddd,J=7.4,7.7,10.9Hz),6.21(1H,d,J=7.6Hz),6.49(1H,d,J=6.2Hz),7.56(1H,d,J=6.2Hz),10.79(1H,d,J=7.6Hz). IwashimaMet al. 13C-NMR(125MHz,CDCl3)dppm14.0,21.3,22.5,27.4,29.0,31.4,36.0,83.5,120.1,129.9,136.0,136.1,150.2,169.5,11.7,192.9. IwashimaMet al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|