LBF20406AM01: Difference between revisions
No edit summary |
No edit summary |
||
| Line 9: | Line 9: | ||
|Common Name=&&N-arachidonoylglycine&& | |Common Name=&&N-arachidonoylglycine&& | ||
|Melting Point=colorless oil [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | |Melting Point=colorless oil [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | ||
|NMR Spectra=^1 H NMR (CDCl3) | |NMR Spectra=^1 H NMR (CDCl3) delta 6.25 (br s 1H), 5.30-5.37 (m, 8H), 4.05 (d, J=5.1Hz, 2H), 2.76-2.82 (m, 6H), 2.22 (t, J=7.8Hz, 2H), 2.04-2.18 (m, 2H), 1.70-1.82 (m, 4H), 1.25-1.35 (m, 6H), 0.89 (t, J=7.1Hz, 3H) [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | ||
|Source= | |Source= | ||
|Chemical Synthesis=This compound was synthesized from arachidonoylchloride and glycine in potassium hydroxide solution. Yield is 34%. [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | |Chemical Synthesis=This compound was synthesized from arachidonoylchloride and glycine in potassium hydroxide solution. Yield is 34%. [[Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659|{{RelationTable/GetFirstAuthor|Reference:Sheskin_T:Hanus_L:Slager_J:Vogel_Z:Mechoulam_R:,J. Med. Chem.,1997,40,659}}]] | ||
Revision as of 23:00, 19 February 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR7017 |
| LipidMaps | LMFA08020003 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20406AM01 |
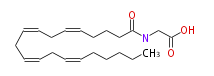
| N-arachidonoylglycine | |
|---|---|
| |
| Structural Information | |
| N-arachidonoylglycine | |
| |
| Formula | C22H35NO3 |
| Exact Mass | 361.261693991 |
| Average Mass | 361.51824 |
| SMILES | C(CCCC=CCC=CCC=CCC=CCCCCC)(NCC(O)=O)=O |
| Physicochemical Information | |
| colorless oil Sheskin_T et al. | |
| This compound was synthesized from arachidonoylchloride and glycine in potassium hydroxide solution. Yield is 34%. Sheskin_T et al. | |
| Binding of this compound to the brain cannabinoid receptor (CBl),Ki(nM)>10000 Sheskin_T et al. | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | 1H NMR (CDCl3) δ 6.25 (br s 1H), 5.30-5.37 (m, 8H), 4.05 (d, J=5.1Hz, 2H), 2.76-2.82 (m, 6H), 2.22 (t, J=7.8Hz, 2H), 2.04-2.18 (m, 2H), 1.70-1.82 (m, 4H), 1.25-1.35 (m, 6H), 0.89 (t, J=7.1Hz, 3H) SheskinTet al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|