LBF20406LT05: Difference between revisions
No edit summary |
No edit summary |
||
| Line 7: | Line 7: | ||
|LipidMaps=LMFA03020012 | |LipidMaps=LMFA03020012 | ||
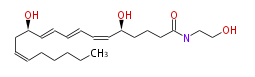
|SysName=N- (2-Hydroxyethyl) - (5S,12R) -dihydroxy- (6-cis,8-trans,10-trans,14-cis) -icosatetraenamide | |SysName=N- (2-Hydroxyethyl) - (5S,12R) -dihydroxy- (6-cis,8-trans,10-trans,14-cis) -icosatetraenamide | ||
|Common Name=&&Leukotriene | |Common Name=&&Leukotriene B_4 ethanolamide&&N- (2-Hydroxyethyl) - (5S,12R) -dihydroxy- (6Z,8E,10E,14Z) -eicosatetraenamide&& | ||
|Source= | |Source= | ||
|Chemical Synthesis= | |Chemical Synthesis= | ||
Revision as of 09:39, 2 June 2010
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR3114 |
| LipidMaps | LMFA03020012 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20406LT05 |
| Leukotriene B4ethanolamide | |
|---|---|
| |
| Structural Information | |
| N- (2-Hydroxyethyl) - (5S,12R) -dihydroxy- (6-cis,8-trans,10-trans,14-cis) -icosatetraenamide | |
| |
| Formula | C22H37NO4 |
| Exact Mass | 379.27225867699997 |
| Average Mass | 379.53352 |
| SMILES | C(=CC[C@@H](O)C=CC=CC=C[C@H](CCCC(=O)NCCO)O)CCCCC |
| Physicochemical Information | |
| LTB4-EA is reported as a potent antagonist about 3 fold higher affinity for the human LTB4 receptor that LTB4. And LTB4-EA antagonizes the LTB4-induced contractions of guinea pig lung parenchyma with 10 nM as an EC50. | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|