LBF18304HP03: Difference between revisions
No edit summary |
No edit summary |
||
| Line 5: | Line 5: | ||
|LipidMaps=LMFA01040046 | |LipidMaps=LMFA01040046 | ||
|SysName=Methyl-13,16-Dihydroperoxy-9,11,14-Octadecatrienoate | |SysName=Methyl-13,16-Dihydroperoxy-9,11,14-Octadecatrienoate | ||
|Mass Spectra=GC-EI-MS(after reduction and TMS-derivatization) | |Mass Spectra=GC-EI-MS(after reduction and TMS-derivatization)[[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780}}]]: m/e=453[M-CH3]; 439[M -CH2CH3]; 437[M-OCH3]; 378[M-HOTMS]; 143[(CH2)6C00CH3], GC-EI-MS(after reduction, hydrogenation and TMS-derivatization)(102) m/e=443[M-OCH3]; 315[SMTO=CH(CH2)11COOCH3] | ||
|UV Spectra=Conjugated diene: <FONT FACE="Symbol">l</FONT>max=233-235nm | |UV Spectra=Conjugated diene: <FONT FACE="Symbol">l</FONT>max=233-235nm[[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1981,16,439|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1981,16,439}}]][[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780}}]] | ||
|IR Spectra=OOH group: 3712-3140cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[bonded], 3530-3510cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[free]; olefinic protons: 3005cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>; conjugated cis, trans diene: 988-979cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>, 953-935cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>; isolated trans unsaturation: 968-960cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP> | |IR Spectra=OOH group: 3712-3140cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[bonded], 3530-3510cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[free]; olefinic protons: 3005cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>; conjugated cis, trans diene: 988-979cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>, 953-935cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>; isolated trans unsaturation: 968-960cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP>[[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1981,16,439|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1981,16,439}}]][[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780}}]] | ||
|NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR | |NMR Spectra=<SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR[[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1981,16,439|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1981,16,439}}]][[Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780|{{RelationTable/GetFirstAuthor|Reference:Neff_WE:Frankel_EN:Weisleder_D:,Lipids,1982,17,780}}]]: C2: 2.3ppm; C8, 17: 1.8-2.2ppm; C9, 10, 11, 12, 14, 15: 5.26-6.28ppm, C13, 16: 4.40-4.42ppm; C18: 1.02ppm; OOH: 8.07ppm | ||
}} | }} | ||
Revision as of 09:00, 12 December 2008
| IDs and Links | |
|---|---|
| LipidBank | DFA8062 |
| LipidMaps | LMFA01040046 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18304HP03 |
| GlcNAca/b1-3Xyla-4Galb1-3GalNAca1-4(NeuAc?1-2NeuGc4Mea1-3)GalNAcb1-4(EtnP-6)GlcNAcb1-3Manb1-4Glcb1-1Cer | |
|---|---|
| |
| Structural Information | |
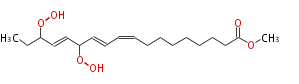
| Methyl-13,16-Dihydroperoxy-9,11,14-Octadecatrienoate | |
| Formula | C19H32O6 |
| Exact Mass | 356.219888756 |
| Average Mass | 356.45378 |
| SMILES | COC(=O)CCCCCCCC=CC=CC(OO)C=CC(CC)OO |
| Physicochemical Information | |
| Spectral Information | |
| Mass Spectra | GC-EI-MS(after reduction and TMS-derivatization) Neff_WE et al.: m/e=453[M-CH3]; 439[M -CH2CH3]; 437[M-OCH3]; 378[M-HOTMS]; 143[(CH2)6C00CH3], GC-EI-MS(after reduction, hydrogenation and TMS-derivatization)(102) m/e=443[M-OCH3]; 315[SMTO=CH(CH2)11COOCH3] |
| UV Spectra | Conjugated diene: lmax=233-235nm Neff_WE et al. Neff_WE et al. |
| IR Spectra | OOH group: 3712-3140cm-1[bonded], 3530-3510cm-1[free]; olefinic protons: 3005cm-1; conjugated cis, trans diene: 988-979cm-1, 953-935cm-1; isolated trans unsaturation: 968-960cm-1 Neff_WE et al. Neff_WE et al. |
| NMR Spectra | 1H-NMR Neff_WE et al. Neff_WE et al.: C2: 2.3ppm; C8, 17: 1.8-2.2ppm; C9, 10, 11, 12, 14, 15: 5.26-6.28ppm, C13, 16: 4.40-4.42ppm; C18: 1.02ppm; OOH: 8.07ppm |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|