LBF20306HX04: Difference between revisions
No edit summary |
No edit summary |
||
| Line 6: | Line 6: | ||
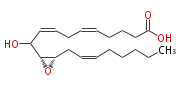
|SysName=10-Hydroxy-11 (R) ,12 (S) -epoxyeicosa-5,8,14 (Z,Z,Z) -trienoic acid | |SysName=10-Hydroxy-11 (R) ,12 (S) -epoxyeicosa-5,8,14 (Z,Z,Z) -trienoic acid | ||
|Common Name=&&HEPOXILIN B3&& | |Common Name=&&HEPOXILIN B3&& | ||
|Reflactive=ACETATE, METHYL ESTER ; [<FONT FACE="Symbol">a</FONT>]X<sub>D</sub><sup>23</sup>=-10.9°(C=0.11, CHLOROFORM) | |Reflactive=ACETATE, METHYL ESTER ; [<FONT FACE="Symbol">a</FONT>]X<sub>D</sub><sup>23</sup>=-10.9°(C=0.11, CHLOROFORM) [[Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126|{{RelationTable/GetFirstAuthor|Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126}}]] | ||
|Solubility=DIETHYL ETHER | |Solubility=DIETHYL ETHER [[Reference:Pace-Asciak_CR:Mizuno_K:Yamamoto_S:,Prostaglandins,1983,25,79|{{RelationTable/GetFirstAuthor|Reference:Pace-Asciak_CR:Mizuno_K:Yamamoto_S:,Prostaglandins,1983,25,79}}]] | ||
|Mass Spectra=METHYL ESTER TMS ETHER ; m/e 311, 282, 269(base peak) | |Mass Spectra=METHYL ESTER TMS ETHER ; m/e 311, 282, 269(base peak) [[Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126|{{RelationTable/GetFirstAuthor|Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126}}]] | ||
|IR Spectra=ACETATE METHYL ESTER ; <FONT FACE="Symbol">n</FONT>(CHLOROFORM) 2956, 1743, 1550, 1372, 1234, 1033, 999cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP> | |IR Spectra=ACETATE METHYL ESTER ; <FONT FACE="Symbol">n</FONT>(CHLOROFORM) 2956, 1743, 1550, 1372, 1234, 1033, 999cm<SUP><FONT SIZE=-1>-</FONT></SUP><SUP><FONT SIZE=-1>1</FONT></SUP> [[Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126|{{RelationTable/GetFirstAuthor|Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126}}]] | ||
|NMR Spectra=ACETATE METHYL ESTER ; <SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(C<SUB><FONT SIZE=-1>6</FONT></SUB>D<SUB><FONT SIZE=-1>6</FONT></SUB>) : <FONT FACE="Symbol">d</FONT> 5.67(dd, J=9.2, 6.4Hz, 1H, 10-CH), 5.52, 5.46, 5.42, 5.35, 5.32, 3.36(s, 3H, OCH<SUB><FONT SIZE=-1>3</FONT></SUB>), 2.95(m, 2H, 7-CH), 2.92(m, 1H, 11-CH), 2.86(ddd, J=7.4, 7.4, 2.1Hz, 1H, 12-CH), 2.30(ddd, J=14.8, 7.4, 7.4Hz, 1H, 13-CH), 2.18(ddd, J=14.8, 7.4, 7.4Hz, 1H, 13-CH), 2.12(t, J=7.4Hz, 2H, 2-CH), 1.98(dt, J=7.4, 7.4Hz, 2H, 4-CH), 1.92(dt, J=8.8, 8.8Hz, 2H, 16-CH), 1.65(s, 3H, COCH<SUB><FONT SIZE=-1>3</FONT></SUB>), 1.60(tt, J=7.4, 7.4Hz, 2H, 3-CH), 1.25(m, 6H), 0.88(t, J=7.0Hz, 3H, 20-CH). | |NMR Spectra=ACETATE METHYL ESTER ; <SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(C<SUB><FONT SIZE=-1>6</FONT></SUB>D<SUB><FONT SIZE=-1>6</FONT></SUB>) : <FONT FACE="Symbol">d</FONT> 5.67(dd, J=9.2, 6.4Hz, 1H, 10-CH), 5.52, 5.46, 5.42, 5.35, 5.32, 3.36(s, 3H, OCH<SUB><FONT SIZE=-1>3</FONT></SUB>), 2.95(m, 2H, 7-CH), 2.92(m, 1H, 11-CH), 2.86(ddd, J=7.4, 7.4, 2.1Hz, 1H, 12-CH), 2.30(ddd, J=14.8, 7.4, 7.4Hz, 1H, 13-CH), 2.18(ddd, J=14.8, 7.4, 7.4Hz, 1H, 13-CH), 2.12(t, J=7.4Hz, 2H, 2-CH), 1.98(dt, J=7.4, 7.4Hz, 2H, 4-CH), 1.92(dt, J=8.8, 8.8Hz, 2H, 16-CH), 1.65(s, 3H, COCH<SUB><FONT SIZE=-1>3</FONT></SUB>), 1.60(tt, J=7.4, 7.4Hz, 2H, 3-CH), 1.25(m, 6H), 0.88(t, J=7.0Hz, 3H, 20-CH). [[Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126|{{RelationTable/GetFirstAuthor|Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126}}]] <SUP><FONT SIZE=-1>1</FONT></SUP><SUP><FONT SIZE=-1>3</FONT></SUP>NMR(C<SUB><FONT SIZE=-1>6</FONT></SUB>D<SUB><FONT SIZE=-1>6</FONT></SUB>) : 134.22, 133.35, 130.17, 127.74, 124.31, 123.37, 70.86, 58.30, 55.71, 50.94, 33.30, 31.71, 29.66, 29.52, 27.59, 26.73, 25.00, 22.88, 20.53, 14.23 [[Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126|{{RelationTable/GetFirstAuthor|Reference:Moghaddam_MF:Gerwick_WH:Ballantine_DL:,J. Biol. Chem.,1990,265,6126}}]] | ||
}} | }} | ||
Revision as of 09:00, 12 December 2008
| IDs and Links | |
|---|---|
| LipidBank | XPR5101 |
| LipidMaps | LMFA03090003 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20306HX04 |
| HEPOXILIN B3 | |
|---|---|
| |
| Structural Information | |
| 10-Hydroxy-11 (R) ,12 (S) -epoxyeicosa-5,8,14 (Z,Z,Z) -trienoic acid | |
| |
| Formula | C20H32O4 |
| Exact Mass | 336.23005951199997 |
| Average Mass | 336.46567999999996 |
| SMILES | C(CC=CC[C@@H]([C@@H](C(O)C=CCC=CCCCC(O)=O)1)O1)CCC |
| Physicochemical Information | |
| DIETHYL ETHER Pace-Asciak_CR et al. | |
| Spectral Information | |
| Mass Spectra | METHYL ESTER TMS ETHER ; m/e 311, 282, 269(base peak) Moghaddam_MF et al. |
| UV Spectra | |
| IR Spectra | ACETATE METHYL ESTER ; n(CHLOROFORM) 2956, 1743, 1550, 1372, 1234, 1033, 999cm-1 Moghaddam_MF et al. |
| NMR Spectra | ACETATE METHYL ESTER ; 1H-NMR(C6D6) : d 5.67(dd, J=9.2, 6.4Hz, 1H, 10-CH), 5.52, 5.46, 5.42, 5.35, 5.32, 3.36(s, 3H, OCH3), 2.95(m, 2H, 7-CH), 2.92(m, 1H, 11-CH), 2.86(ddd, J=7.4, 7.4, 2.1Hz, 1H, 12-CH), 2.30(ddd, J=14.8, 7.4, 7.4Hz, 1H, 13-CH), 2.18(ddd, J=14.8, 7.4, 7.4Hz, 1H, 13-CH), 2.12(t, J=7.4Hz, 2H, 2-CH), 1.98(dt, J=7.4, 7.4Hz, 2H, 4-CH), 1.92(dt, J=8.8, 8.8Hz, 2H, 16-CH), 1.65(s, 3H, COCH3), 1.60(tt, J=7.4, 7.4Hz, 2H, 3-CH), 1.25(m, 6H), 0.88(t, J=7.0Hz, 3H, 20-CH). Moghaddam_MF et al. 13NMR(C6D6) : 134.22, 133.35, 130.17, 127.74, 124.31, 123.37, 70.86, 58.30, 55.71, 50.94, 33.30, 31.71, 29.66, 29.52, 27.59, 26.73, 25.00, 22.88, 20.53, 14.23 Moghaddam_MF et al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|