LBF20407HO07: Difference between revisions
No edit summary |
No edit summary |
||
| Line 13: | Line 13: | ||
|NMR Spectra=METHYL ESTER ; <SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(90MHz) : <FONT FACE="Symbol">d</FONT> 6.56(dd, J=11, 14.5Hz, 1H, 13-CH), 6.02(dd, J=11,11Hz, 1H, 12-CH), 5.7(dd, J=6.8, 14.5Hz, 1H,14-CH), 5.51(dd, J=6,11Hz, 1H, 11-CH), 4.16(dd, J=6.8, 6.8Hz, 1H, 15-CH), 3.69(s, 3H, OCH<SUB><FONT SIZE=-1>3</FONT></SUB>), 2.98(2H, 10-CH), 2.84(2H, 7-CH), 2.35(2H, 2-CH), 2.08(2H, 4-CH), 1.47(2H, 16-CH), 2.3-0.9(11H) [[Reference:Baldwin_JE:Davies_DI:Hughes_L:Gutterridge_NJA:,J. Chem. Soc. Perkin I,1979,,115|{{RelationTable/GetFirstAuthor|Reference:Baldwin_JE:Davies_DI:Hughes_L:Gutterridge_NJA:,J. Chem. Soc. Perkin I,1979,,115}}]] | |NMR Spectra=METHYL ESTER ; <SUP><FONT SIZE=-1>1</FONT></SUP>H-NMR(90MHz) : <FONT FACE="Symbol">d</FONT> 6.56(dd, J=11, 14.5Hz, 1H, 13-CH), 6.02(dd, J=11,11Hz, 1H, 12-CH), 5.7(dd, J=6.8, 14.5Hz, 1H,14-CH), 5.51(dd, J=6,11Hz, 1H, 11-CH), 4.16(dd, J=6.8, 6.8Hz, 1H, 15-CH), 3.69(s, 3H, OCH<SUB><FONT SIZE=-1>3</FONT></SUB>), 2.98(2H, 10-CH), 2.84(2H, 7-CH), 2.35(2H, 2-CH), 2.08(2H, 4-CH), 1.47(2H, 16-CH), 2.3-0.9(11H) [[Reference:Baldwin_JE:Davies_DI:Hughes_L:Gutterridge_NJA:,J. Chem. Soc. Perkin I,1979,,115|{{RelationTable/GetFirstAuthor|Reference:Baldwin_JE:Davies_DI:Hughes_L:Gutterridge_NJA:,J. Chem. Soc. Perkin I,1979,,115}}]] | ||
|Source= | |Source= | ||
|Chemical Synthesis=[[Reference:Baldwin_JE:Davies_DI:Hughes_L:Gutterridge_NJA:,J. Chem. Soc. Perkin I,1979,,115|{{RelationTable/GetFirstAuthor|Reference:Baldwin_JE:Davies_DI:Hughes_L:Gutterridge_NJA:,J. Chem. Soc. Perkin I,1979,,115}}]];> {{Image200| | |Chemical Synthesis=[[Reference:Baldwin_JE:Davies_DI:Hughes_L:Gutterridge_NJA:,J. Chem. Soc. Perkin I,1979,,115|{{RelationTable/GetFirstAuthor|Reference:Baldwin_JE:Davies_DI:Hughes_L:Gutterridge_NJA:,J. Chem. Soc. Perkin I,1979,,115}}]];> {{Image200|LBF20407HO07FT0001.gif}} | ||
|Metabolism=When arachidonic acid is oxygenated by 15-lipoxygenase, 15(S)-hydroperoxy-5,8,11,13-(Z,Z,Z,E)-eicosatetraenoic acid is produced [[Reference:Bryant_RW:Bailey_JM:Schewe_T:Rapoport_SM:,J. Biol. Chem.,1982,257,6050|{{RelationTable/GetFirstAuthor|Reference:Bryant_RW:Bailey_JM:Schewe_T:Rapoport_SM:,J. Biol. Chem.,1982,257,6050}}]];>. The latter compound is reduced to a corresponding 15(S)-hydroxy acid with whole cells or crude enzyme preparations. | |Metabolism=When arachidonic acid is oxygenated by 15-lipoxygenase, 15(S)-hydroperoxy-5,8,11,13-(Z,Z,Z,E)-eicosatetraenoic acid is produced [[Reference:Bryant_RW:Bailey_JM:Schewe_T:Rapoport_SM:,J. Biol. Chem.,1982,257,6050|{{RelationTable/GetFirstAuthor|Reference:Bryant_RW:Bailey_JM:Schewe_T:Rapoport_SM:,J. Biol. Chem.,1982,257,6050}}]];>. The latter compound is reduced to a corresponding 15(S)-hydroxy acid with whole cells or crude enzyme preparations. | ||
}} | }} | ||
{{Lipid/Footer}} | {{Lipid/Footer}} | ||
Revision as of 19:00, 25 November 2009
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR6103 |
| LipidMaps | - |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20407HO07 |
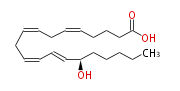
| 15 (S) -Hydroxy-5,8,11,13- (Z,Z,Z,E) -eicosatetraenoic acid | |
|---|---|
| |
| Structural Information | |
| 15 (S) -Hydroxy-5,8,11,13- (Z,Z,Z,E) -eicosatetraenoic acid | |
| |
| Formula | C20H32O3 |
| Exact Mass | 320.23514489 |
| Average Mass | 320.46628 |
| SMILES | C(CC[C@@H](C=CC=CCC=CCC=CCCCC(O)=O)O)CC |
| Physicochemical Information | |
| DIETHYL ETHER Baldwin_JE et al. | |
|
Baldwin_JE et al.;> | |
| When arachidonic acid is oxygenated by 15-lipoxygenase, 15(S)-hydroperoxy-5,8,11,13-(Z,Z,Z,E)-eicosatetraenoic acid is produced Bryant_RW et al.;>. The latter compound is reduced to a corresponding 15(S)-hydroxy acid with whole cells or crude enzyme preparations. | |
| Spectral Information | |
| Mass Spectra | METHYL ESTER TMS ETHER ; m/e 406(M+), 391, 335, 316, 305, 225, 173 Baldwin_JE et al. |
| UV Spectra | METHYL ESTER ; l (ISOOCTANE) = 236nm(e 27200) Baldwin_JE et al. |
| IR Spectra | |
| NMR Spectra | METHYL ESTER ; 1H-NMR(90MHz) : d 6.56(dd, J=11, 14.5Hz, 1H, 13-CH), 6.02(dd, J=11,11Hz, 1H, 12-CH), 5.7(dd, J=6.8, 14.5Hz, 1H,14-CH), 5.51(dd, J=6,11Hz, 1H, 11-CH), 4.16(dd, J=6.8, 6.8Hz, 1H, 15-CH), 3.69(s, 3H, OCH3), 2.98(2H, 10-CH), 2.84(2H, 7-CH), 2.35(2H, 2-CH), 2.08(2H, 4-CH), 1.47(2H, 16-CH), 2.3-0.9(11H) Baldwin_JE et al. |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|