LBF18108HP04
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA8069 |
| LipidMaps | LMFA01040053 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18108HP04 |
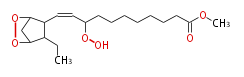
| Methyl-11- (3,5-epidioxy-2-ethylcyclopentyl) -9-hydroperoxy-10-undecenoate | |
|---|---|
| |
| Structural Information | |
| Methyl-11- (3,5-epidioxy-2-ethylcyclopentyl) -9-hydroperoxy-10-undecenoic acid | |
| |
| Formula | C19H32O6 |
| Exact Mass | 356.219888756 |
| Average Mass | 356.45378 |
| SMILES | C(C2CC)(O1)CC(C2C=CC(OO)CCCCCCCC(=O)OC)O1 |
| Physicochemical Information | |
| Photoenhancement of linoleate peroxidation(TypeII) Frankel_EN Frankel_EN Neff_WE et al.. It is produced from a 13-peroxyradical of linoleate via 1,3-cyclization Frankel_EN Frankel_EN Neff_WE et al. Oconnor_DE et al. Oconnor_DE et al.. | |
| It reacts with DNA in the presence of Fe ions and ascorbic acid Fujimoto_K et al.. | |
| Spectral Information | |
| Mass Spectra | EI-MS(after reduction and TMS) Oconnor_DE et al.: m/e=543[M-CH3]; 468[M-HOTMS]; 401[M-(CH2)7COOCH3]; 378[M-2xHOTMS]; 352[468-SMTO=CHCH2]; 323[352-CH2CH3]; 285[CH=CHCH(OTMS)(CH2)7COOCH3]; 259[SMTO=CH(CH2)7COOCH3]; 217[SMTO-CH=CH-CH=OTMS] |
| UV Spectra | |
| IR Spectra | OOH group: 3620-3010cm-1[bonded], 3520cm-1[free]; isolated trans unsaturation: 960cm-1 Neff_WE et al. |
| NMR Spectra | 1H-NMR Oconnor_DE et al. Oconnor_DE et al.: C9: 4.26-4.35; C10: 5.47-5.52; C11: 5.44-6.03; C12: 2.66-2.92; C13: 4.42-4.53; C15: 4.53-4.66; C16: 1.90-2.16; OOH: 7.89-7.94; 13C-NMR Oconnor_DE et al. Oconnor_DE et al.: C9: 86.3-86.6; C10: 129.8-132.4; C11: 132.4-135.0; C12: 45.9-50.8; C13: 79.8-80.5C14: 39.3-44.1; C15: 81.7-83.0; |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|