LBF18303HP04
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA8054 |
| LipidMaps | LMFA01040038 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF18303HP04 |
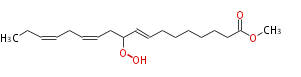
| Methyl-10-hydroperoxy- (8E,12Z,15Z) -octadecatrienoic acid | |
|---|---|
| |
| Structural Information | |
| Methyl-10-hydroperoxy- (trans-8,cis-12,cis-15) -octadecatrienoic acid | |
| |
| Formula | C19H32O4 |
| Exact Mass | 324.23005951199997 |
| Average Mass | 324.45498 |
| SMILES | CCC=CCC=CCC(OO)C=CCCCCCCC(=O)OC |
| Physicochemical Information | |
| Oxidation of linoleate by singlet oxygen Frankel_EN Frankel_EN Frankel_EN . | |
| Pysiological damages are induced by these hydroperoxides which are incorporated into bodies or synthesized endogenously. Logani_MK et al. Sevanian_A et al. Fujimoto_K . It reacts with DNA in the presence of Fe ions and ascorbic acid Fujimoto_K et al.. | |
| Isomerization of hydroperoxides: Positional isomers at 9, 10, 12, 13, 15, and 16 and their geometric isomers of cis, cis, trans- or cis, trans, trans are formed. The proportion of 9-, 16- isomers is higher than that of 12-, 13- isomers. | |
| Spectral Information | |
| Mass Spectra | EI-MS(Me-ester; after reduction and hydrogenation) Chan_HWS : m/e=201[O=CH(CH2)8C(=OH)OCH3]; 172[CH2(CH2)7C(=O)OCH3];169[O=CH(CH2)8C=O]; GC-EI-MS (Me-ester; after reduction, TMS) TeraoJet al.: m/e=271[SMTO=CH-CH=CH(CH2)6COOCH3], GC-EI-MS(Me-ester;after reduction, hydrogenation) |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | ||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|