LBF20406CV06
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | XPR8002 |
| LipidMaps | LMFA03120002 |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20406CV06 |
| Clavulone II | |
|---|---|
| |
| Structural Information | |
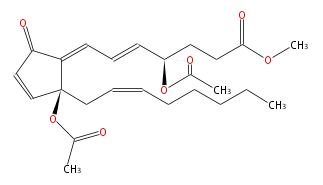
| Methyl-4R- (trans-5,trans-7) -4-acetoxy-7- [ 2S-acetoxy-2- (cis-2-octenyl) -5-oxo-3-cyclopentenylidene] -5-heptenoic acid | |
| |
| Formula | C25H34O7 |
| Exact Mass | 446.230453442 |
| Average Mass | 446.53326000000004 |
| SMILES | O(C(C)=O)[C@@](C1=CC=C[C@@H](CCC(OC)=O)OC(C)=O)(CC=CCCCCC)C=CC1=O |
| Physicochemical Information | |
| [ α ]D +10.9°(C 0.35, CHCl3) KikuchiHet al. | |
| Clavulones are soluble in MeOH, EtOH, CHCl3, or hexane. | |
| Clavulones were isolated from Japanese soft coral, Stolonifer Clavularia viridis Quoy and Gaimard. Kikuchi_H et al. Kobayashi_M et al. Kikuchi_H et al. Kobayashi_M et al. | |
| Clavulone II was synthesized from cyclopentadiene and methyl 3-chloroformylpropionate as a racemic form. Corey_EJ et al.Clavulones were synthesized from L-(+)-diethyl tartarate and D-mannitol as a natural form. Nagaoka_H et al.Other synthesis of clavulone. Hashimoto_S et al. Shibasaki_M et al. Klunder_AJH et al. Takemoto M et al. Zhu_J et al. Takeda_K et al. | |
| The biosynthesis of clavulones is suggested to proceed from arachidonic acid via 8(R)-HEPETE and pre-clavulone A. Corey_EJ Corey_EJ et al. Corey_EJ et al. Corey_EJ et al. | |
| Clavulones showed a significant anti-inflammatory effect at 30 mu g/ml by the fertile egg test. Kikuchi_H et al.Clavulone showed positive chronotropic action on the cultured myocardial cells. Honda_A et al.Clavulone II showed strong antiproliferative activity in the human myeloid leukemia (HL-60) cells (IC_{50} 0.2 mu g/ml). Honda_A et al.Clavulone II was entrapped into lipid microspheres of 0.2 mu m diameter to lipo-drug. Daily treatment with lipo-clavulone II (12.5 mg/kg/day, i.p.) on days 1 through 5 markedly prolonged the survival time (73% ILS) of mice inoculated with sarcoma 180 as compared with that of corresponding dose of respective free clavulone II. Honda_A et al. | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | λ max(EtOH) 230 nm( ε 14500),292 nm( ε 19300) KikuchiHet al. |
| IR Spectra | ν max(film)1730,1700,1640,and 1230cm-1 KikuchiHet al. |
| NMR Spectra | 1H-NMR(270MHz,CDCl3) δ ppm0.88(3H,t,J=6.9Hz),2.07(3H,s),2.08(3H,s),2.38(2H,t,J=7.5Hz),2.69(1H,dd,J=8,14.5Hz),2.88(1H,dd,J=7,14.5Hz),3.68(3H,s),5.22(1H,m),5.52(1H,dt,J=10.9,8Hz),5.42(1H,q,J=7Hz),6.02(1H,dd,J=7,14.5Hz),6.41(1H,d,J=6.3Hz),6.75(1H,dd,J=11.6,14.5Hz),6.87(1H,d,J=11.6Hz),7.47(1H,d,J=6.3Hz). KikuchiHet al.13C-NMR(67.8MHz,CDCl3) δ ppm14.0(q),21.0(q),21.2(q),22.5(t),27.4(t),29.1(t),29.1(t),29.6(t),31.5(t),36.0(t),51.8(q),72.8(d),85.1(s),121.1(d),126.9(d),129.3(d),135.0(d),137.0(s),141.3(d),158.1(d),169.5(s),169.9(s),172.9(s),193.4(s). KikuchiHet al. |
| Other Spectra | CD λ ext(EtOH)( Δ ε )nm 250(+4.2),293(-3.4). KikuchiHet al. |
| Chromatograms | |