LBF20406HP06
| LipidBank Top (トップ) |
Fatty acid (脂肪酸) |
Glycerolipid (グリセロ脂質) |
Sphingolipid (スフィンゴ脂質) |
Journals (雑誌一覧) |
How to edit (ページの書き方) |
| IDs and Links | |
|---|---|
| LipidBank | DFA8085 |
| LipidMaps | - |
| CAS | |
| KEGG | {{{KEGG}}} |
| KNApSAcK | {{{KNApSAcK}}} |
| mol | LBF20406HP06 |
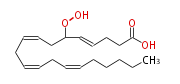
| 6-Hydroperoxy- (4E,8Z,11Z,14Z) -eicosatetraenoic acid | |
|---|---|
| |
| Structural Information | |
| 6-Hydroperoxy- (trans-4,cis-8,cis-11,cis-14) -eicosatetraenoic acid | |
| |
| Formula | C20H32O4 |
| Exact Mass | 336.23005951199997 |
| Average Mass | 336.46567999999996 |
| SMILES | C(CC=CCC=CCC=CCC(OO)C=CCCC(O)=O)CCC |
| Physicochemical Information | |
| Oxidation of arachidonic acid by singlet-oxygen Terao_J et al. Porter_NA et al.. | |
| Isomerization of hydroperoxides : Both 5- and 15-hydroperoxide were generated predominantly by autooxidation or singlet-oxygen mediated oxidation Terao_J et al. Yamagata_S et al. Peers_KE et al.. However, the proportion of each isomer generated become almost equal by supplementation of hydrogen donor such as tocopherol Porter_NA et al. Terao_J et al. Peers_KE et al.. | |
| Spectral Information | |
| Mass Spectra | GC-EI-MS(Me-ester; after reduction and TMS) TeraoJet al.: m/e=406[M]; 316[M-HOTMS]; GC-EI-MS(Me-ester; after reduction and TBDMS) TeraoJet al.: m/e=448[M], 391[M-(CH3)3C], GC-EI-MS(Me-ester; after reduction, hydrogenation and TMS) TeraoJet al. Porter_NA et al.: m/e=399[M-CH3] |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | |
| Other Spectra | |
| Chromatograms | |
| Reported Metabolites, References | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|